A certain injury pathway in neurons may cause the loss of synapses in diseases like Alzheimer’s and ALS.
This pathway, called DLK, has received recent attention as a candidate drug target because it contributes to the deterioration of damaged neurons. The new findings further expand that interest by suggesting that inhibiting DLK may help neurons to maintain working synapses, which is more useful than simply preventing damaged neurons from dying.
The pathway, studied here in fruit flies, is similar to the pathway in the neurons of mammals and humans. The research team, who published their results in the journal eLife, found a new relationship between the injury pathway and a protein in neurons called kinesin—specifically, a kinesin called Unc-104.
When neurons communicate with each other, they extend a projection called an axon toward each other and form sites for information exchange called synapses. Transmitting information between synapses requires many protein molecules, which are manufactured within the neuron cell body and ferried over long distances within axons by kinesins such as Unc-104. The Unc-104 kinesin seems to carry many of the proteins needed for the neuron to release neurotransmitters for information exchange.
The researchers found that the injury pathway is triggered when Unc-104 is damaged or mutated. This builds upon previous knowledge that the pathway is required for the key responses neurons make when damaged, including initiating the neuron to repair itself or die, depending on the context.
When Unc-104 is damaged or mutated, synapses become defective. Previously, researchers assumed that this is because of a failure to transport synaptic proteins to synapse sites. However, in their study, the researchers found that turning off the neuronal injury pathway can restore the function of these mutant synapses.
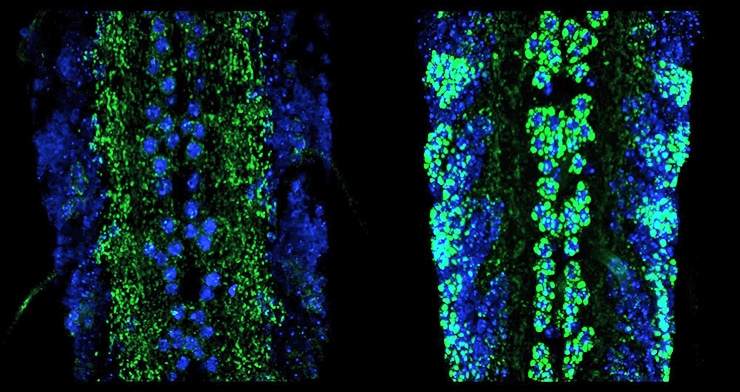
To study the role of the kinesin and the injury pathway in the malfunction of synapses, the researchers imaged the synapses of dissected fruit fly larvae using a confocal microscope. Li also used electrophysiology to measure how well the synapses were firing. The team found that when they shut down the injury pathway, the function of that synapse returned.
“That was really striking,” says Catherine Collins, an associate professor of molecular, cellular, and developmental biology at the University of Michigan. “It told us that the axon injury pathway was causing these major problems in synapses.”
The team found that the pathway becomes activated when the Unc-104 kinesin is impaired, and that once activated, the pathway shuts down the formation of many of the synaptic proteins which are normally transported in axons.
How motor neurons falter in spinal muscular atrophy
“In the fruit fly system, we know the neuroanatomy really well. We can actually see how impairment of kinesin is affecting things in cell bodies, synapses and neurons,” Collins says. “When we turned down the injury pathway in the kinesin mutants, we could see a huge mass of synaptic proteins accumulating in the cell body. That led us to the idea that the injury pathway turns down the levels of many synaptic proteins, leading to synapse malfunction.”
The findings imply that activation of the injury pathway has negative consequences for synapses. Complementing this work, recent findings published in Science Translational Medicine suggest that the injury pathway may be activated in patients with neurodegenerative diseases ALS and Alzheimer’s disease.
Restricting the pathway can also delay symptoms of these diseases in mouse models. This draws attention to the pathway as a potential therapeutic target.
But this could also hinder beneficial functions of the injury pathway, Collins says.
“When the injury pathway was knocked down in flies, the massive accumulations of proteins in cell bodies suggested to us that it functions as a stress response mechanism, to prevent unwanted build-up of proteins when axonal transport is impaired,” she says.
Injection sends ‘genetic cargo’ to neurons all over the body
Next, the researchers want to understand why the injury pathway appears specifically tuned to Unc-104 for its activation, and how the pathway reduces synaptic proteins.
Source: University of Michigan



