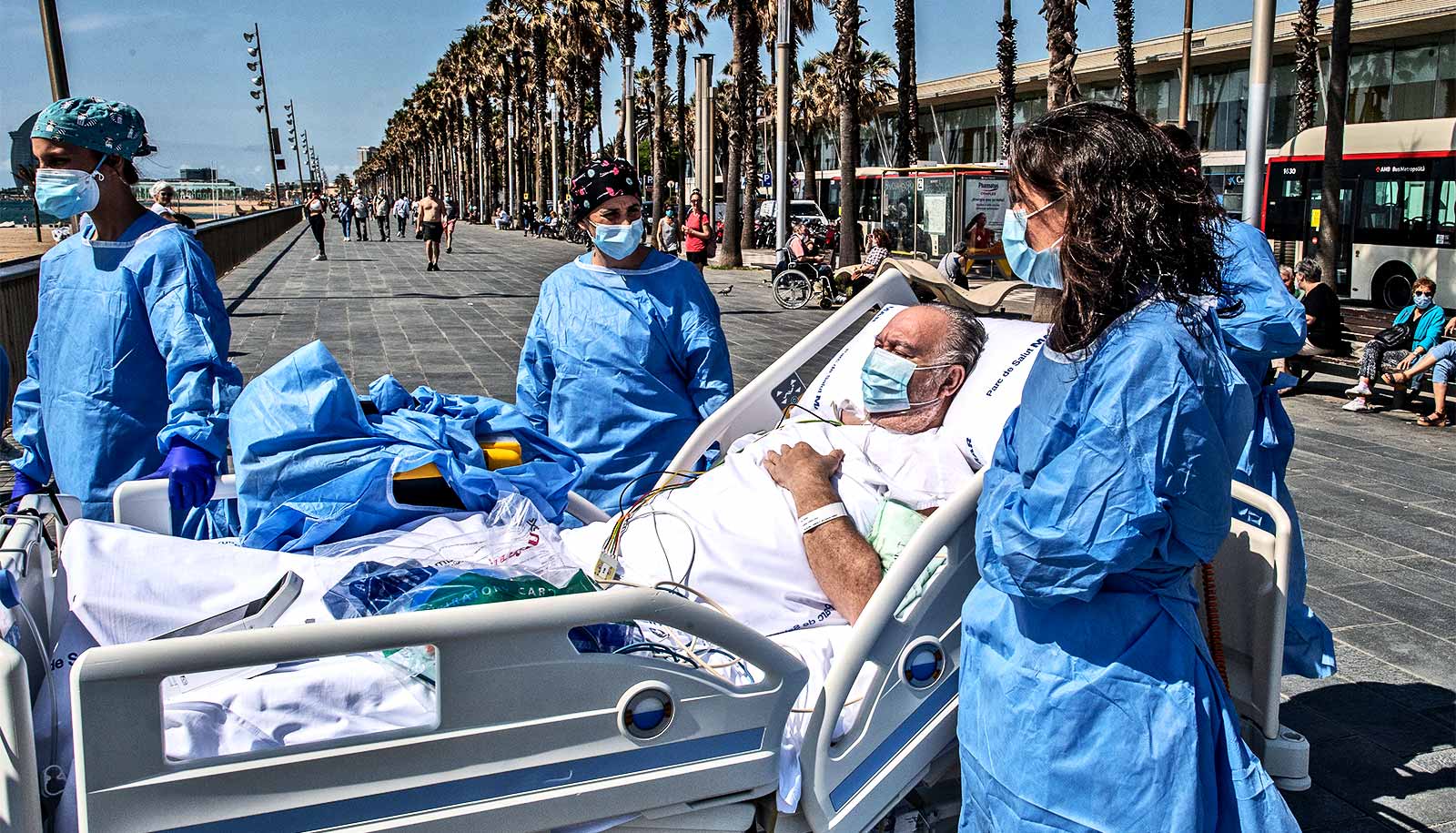
For some individuals with COVID-19, recovering from the acute phase of the infection is only the beginning, John Swartzberg warns.
Worrying reports now indicate that the coronarvirus may be capable of inflicting long-lasting damage to the lungs, heart, and nervous system, and researchers are closely watching to see if the kidneys, liver, and gastrointestinal tract may be susceptible to persistent damage as well.
Some patients also report symptoms that remain weeks, even months, after becoming infected, leading some to suspect that the virus may be able to spark conditions like chronic fatigue syndrome.
“In the beginning, our model for understanding this infection was to treat it like another respiratory virus like influenza,” says Swartzberg, clinical professor emeritus of infectious diseases and vaccinology in the University of California, Berkeley-UC San Francisco Joint Medical Program.
“I think one of the most unfortunate and interesting things about this virus is that its interaction with us is actually far more complicated than that.”
Here, Swartzberg explains what we know about the virus’s potential to inflict long-term damage to the body and how patients can lessen their risk of complications. He also cautions, however, that after only about six months of the SARS-CoV-2 virus, it’s too early to say if any persistent effects are chronic or long-term, or if we’ve identified all the possible long-term impacts:
What are some of the persistent or long-lasting health complications that doctors are seeing in patients who have recovered from acute COVID-19?
One thing we didn’t anticipate was that the virus seems to accelerate a great deal of scarring in the lungs. And if lung tissue is replaced with scar tissue, it is no longer functional as regular pulmonary tissue, which translates to poor gas exchange. What we really fear is long-term shortness of breath that could extend anywhere from being very mild to severely limiting. There is also a disturbing report looking at computerized tomography (CT) scans of asymptomatic people that found they were left with some scar tissue. So, this could even be happening on a subclinical level.
Another area is the heart. There is evidence now that the virus can directly attack heart muscle cells, and there’s also evidence that the cytokine storm that the virus triggers in the body not only damages the lungs, but can damage the heart. We don’t know what the long-term effects of that may be, but it could be that we will have a population of people who survive COVID-19 only to go on and have chronic cardiac problems.
The third organ system that we’re now pretty clear about is the central nervous system. There is evidence of direct involvement of the virus with neurons, and also the cytokine storm and inflammatory mediators can cause damage to the central nervous system. This is manifesting itself not only in neurologic clinical findings, but also psychological findings. We’re seeing patients post-discharge struggling with psychological challenges, almost like PTSD. And, we’re also seeing some cognitive defects in some people that are very disturbing.
We’ve also seen damage to the kidneys from the cytokines, and there is also evidence that the virus can bind to receptors in the liver, though we haven’t seen significant liver disease yet in patients. Finally, the gastrointestinal tract itself has virus receptors, and about 15% of people, especially children, present with gastrointestinal symptoms. But, so far, there is no evidence that this will cause persistent symptoms.
Finally, it has become clear that infection with SARS-CoV-2 triggers abnormal clotting of the blood in some people. This has led to pulmonary emboli, which are blood clots that travel to and damage the lungs, and strokes, which are blood clots in the vascular system of the brain. Both pulmonary emboli and strokes may have long-term consequences for these two organs.
Then, in pediatrics, there’s this multi-system inflammatory syndrome in children, which appears to occur not typically with acute infection, but following the acute infection by a short period of a few weeks. This is where multiple systems are involved with inflammation, including skin, joints, kidneys, lungs, and heart. And some of these kids can be very sick, with rare deaths.
I think I’ve gone through almost all the organ systems, and the ones that I think are highly likely to suffer persistent complications are lungs, the heart and maybe central nervous system. But, the rate at which we are learning is enormously fast.
I’m sure if you come back to me in three or six months, the list will be longer in some places, but maybe we will have eliminated some potential chronic problems.
Do scientists understand why the SARS-CoV-2 virus is triggering all these potentially chronic health complications, when other viruses like the common cold or influenza rarely do?
This is probably one of the key questions for immunologists. There are myriad hypotheses, but no direct answers yet. We’re still dancing with that question.
Do we know if any populations are more or less at risk of having persistent complications from COVID-19?
We don’t know the answer to that yet. We do know that people from lower socio-economic groups in our society have more severe disease and have higher mortality. A sufficient explanation for that is social determinants of health. There’s no evidence that race has an impact on the rates of either acute infection or persistent complications, beyond what could be explained by the fact that resource-challenged populations have many more risk factors for serious disease, including hypertension, diabetes, and obesity, and have less access to care. I think we’ll likely see more long-term consequences in those populations, too, because they have more severe disease.
There are some interesting observations that people with blood group A may be more predisposed to severe disease. That’s still a hypothesis, but it seems to have held up over about two or three months, which is a long time.
I’ve read about COVID-19 “long-haulers,” who are often young or middle-aged adults who get sick with the virus and are still sick months later. What do we know about these cases?
We’re seeing people with chronic fatigue syndrome, which is hard to study because we don’t have clearly defined biomarkers for it, but we just don’t understand at this point whether that’s more common in younger people or not.
I think a question mark remains as to how many persistent problems we’ll see in people who are infected, but didn’t get that sick. The people who are getting looked at most carefully are people who have been very sick and hospitalized. People who are sick, but not needing hospitalization, aren’t studied as much, and people who are asymptomatically infected are hardly studied at all.
The emergence of more problems in younger people is also difficult to know. Clearly, in the month of June, the average age of people infected (trended) downward. The reasons for that are still debated, but I do think a major reason is that younger people are generally less likely to follow (social distancing) recommendations than older people. I don’t know if that’s a sufficient explanation, but I think it is an important one.
Are there any steps people can take to now reduce their risk of developing persistent complications, if they do contract COVID-19 in the future?
If it’s true, which it appears to be at this point, that the people who get the most sick are the most likely to have persistent problems, then the question should be: What can you do to prevent getting really sick? And the answer is, look at the risk factors that are modifiable. Age is not a modifiable risk factor. But diabetes, hypertension, and obesity are. Anything that is causing chronic inflammation in the body. Smoking, including vaping, could all be under that framework. So, I think that the answer to that is to optimize your health.
Source: UC Berkeley