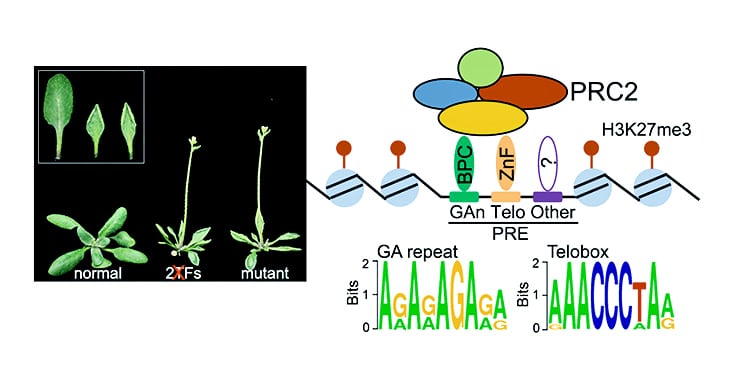
Biologists have identified small sequences in plant DNA that act as “signposts” for shutting off gene activity, directing the placement of proteins that silence gene expression.
Manipulating these short DNA fragments offers the potential to grow plants with enhanced activation of certain traits, such as fruiting or seed production. The finding may also have implications for understanding gene regulation in both plants and animals.
Turning genes on or off
A plant has one genome, a specific sequence of millions of basepairs of nucleotides. Yet how this genome is expressed can vary from cell to cell, and it can change as a plant goes through various life stages, from germination to vegetative growth to flowering to dormancy. Some genes must be turned on and others shut off to ensure each plant cell is doing what it needs to do when it needs to do it.
“Part of identity is what you aren’t,” says Doris Wagner, senior author on the study and a professor in the University of Pennsylvania’s biology department.
“Especially for plants because they are so changeable and susceptible to environmental conditions, the part of the genome that is not needed, or that might be providing exactly the wrong information, needs to be shut off reliably in each condition. This information is then passed on to daughter cells,” Wagner explains.
“With these short sequences,” she says, “we could try to manipulate them using gene-editing techniques to alter gene expression without adding any foreign genetic material to the plant and epigenetically alter expression of traits.”
Polycomb repression
The study focused on the form of gene regulation known as Polycomb repression. Polycomb protein complexes were first discovered in fruit flies, shown to tightly compact DNA and represent an epigenetic modification leading to gene silencing.
Polycomb complexes were later discovered in plants and mammals. In all species, they play important roles in determining cell identity, helping plant cells remember, for example, that they are leaf cells or flower cells.
Despite some studies implicating short segments of DNA called Polycomb response elements, or PREs, in the Polycomb targeting process in flies, questions remained about whether such PREs played a broad role in gene silencing in mammals or plants.
Wagner’s team examined the Polycomb complex called PRC2. Using large-scale datasets collected by her lab and others, the researchers identified 170 segments of DNA in the plant species Arabidopsis thaliana that were likely to be PREs. Testing five of these candidate PREs, they confirmed that they acted just as PREs did in fruit flies, recruiting the Polycomb complex to specific parts of the plant genome.
The researchers then identified 55 transcription factors, proteins that bind specific DNA sequences and help regulate how DNA is turned into RNA, that strongly bound to the PREs, and they verified that 30 of them physically interacted with PRC2.
“This is just what a recruiter should do,” Wagner says, “find the appropriate region in the genome and bring in Polycomb.”
Wanting to know more about what elements in the DNA sequence itself marked it for targeting by Polycomb complexes, the researchers went back to the 170 PRE candidates, computationally identifying short DNA sequences called cis motifs, which are what transcription factors recognize when they scan the genome for their target genes.
How plants slow their growth under stress
With additional analysis, Wagner and colleagues found two of the cis motifs matched up with two of the previously identified transcription factors. Putting these cis motifs into a plant cell genome revealed they were sufficient for recruiting Polycomb, making them essentially a synthetic PRE.
“We brought together the cis (within DNA sequence) and trans (acting on a DNA sequence) factors to show how Polycomb targets specific PREs and broadly regulates plant gene expression,” Wagner says.
“This is the first demonstration that this mechanism—recruitment of Polycomb by these signposts in the DNA—acts in species outside of fruit flies. In the future, I could use these motifs to epigenetically enhance desirable traits such as yield or drought tolerance without significantly changing the coding sequence,” she says.
Future plans and implications
In follow-up work, Wagner wants to explore PREs and these motifs and transcription factors in plant species besides Arabidopsis. She’d also like to investigate how rapidly the system can change if, for example, a plant is exposed to water or salt stress.
The findings may also guide the work of researchers outside of the plant field, notes Kenneth Zaret, director of the university’s Institute of Regenerative Medicine, who did not participate in the current study but who studies gene regulation in animals.
Can a dose of probiotics drought-proof plants?
“Finding specific DNA sequences that mediate the action of the Polycomb repressive system has been the Holy Grail for mammalian cell biologists,” Zaret says. “The rigorous approach of the Wagner study beautifully revealed a mechanism of repression of gene activity that will no doubt have implications beyond the plant world.”
The research paper appears in the journal Nature Genetics.
Additional researchers who contributed to the work are from the University of Pennsylvania; the University of Georgia; CSIRO Agriculture and Food; the University of California, San Diego; the Scripps Research Institute; the Hebei Normal University in China; the University of Edinburgh; and Western University. National Science Foundation grants, National Institutes of Health grants, and other sources provided support for the study.
Source: University of Pennsylvania