A new drug delivery system, called cavitation dose painting, would make getting drugs where they need to go more like “paint by numbers” and less like paintball.
If the drug needs to enter the bloodstream and circulate throughout your body for treating disease wherever it may be, the paintball-like delivery system may work, but it won’t work for targeted and precise drug delivery.
A technique more like “painting by numbers,” would allow precise delivery of a certain amount of drugs to an exact location. Researchers at the McKelvey School of Engineering and the School of Medicine at Washington University in St. Louis are developing the tools necessary for such a system. Their research appears in Scientific Reports.
Microbubbles in action
Using focused ultrasound with its contrast agent, microbubbles, to deliver drugs across the blood-brain barrier (FUS-BBBD), the research team, led by Hong Chen, assistant professor of biomedical engineering at McKelvey School of Engineering, and assistant professor of radiation oncology at the School of Medicine, was able to overcome some of the uncertainty of drug delivery.
This method takes advantage of the microbubbles expanding and contracting when they interact with the ultrasound, essentially pumping the intravenously-delivered drug to wherever the ultrasound is pointing.
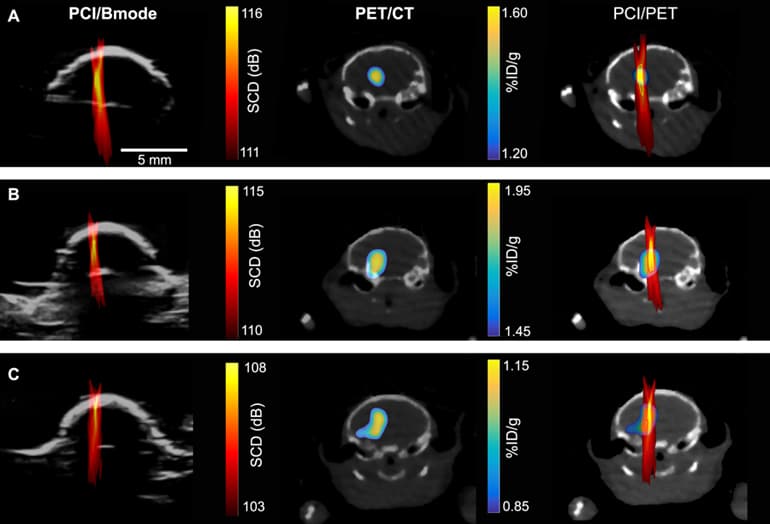
To determine where and how much of the drugs were being delivered, the researchers used nanoparticles tagged with radio labels to represent drug particles, then used positron emission tomography (PET) imaging to track their whereabouts and concentrations. They could then create a detailed image, showing where the nanoparticles were going and in what concentrations.
“The problem is, PET imaging is expensive and associated with radioactive exposure,” Chen says.
So the team turned to passive cavitation imaging (PCI), an ultrasound imaging technique that has been under development by several groups for imaging the spatial distribution of microbubble cavitation, or the oscillation of microbubbles in the ultrasound field.
To determine whether PCI could also accurately determine the amount of drugs at a certain location, they correlated a PCI image with a PET image (which they knew can quantify the concentration of radioactive agents).
“We found there’s pixel by pixel correlation between the ultrasound imaging and the PET imaging,” says Yaoheng Yang, the lead author of the study and a second-year PhD student in the biomedical engineering department. The PCI image, therefore, can predict where a drug goes and how much drug is there. Hence, she called the new technique cavitation dose painting.
Brain drug delivery
Going forward, Chen believes this method could drastically change the way some drugs are delivered. Using cavitation dose painting in tandem with focused ultrasound will allow doctors to deliver precise amounts of drugs to specific locations, for example, targeting different areas of a tumor with exactitude.
“I think this cavitation dose painting technique in combination with focused ultrasound-enabled brain drug delivery opens new horizons in spatially targeted and modulated brain drug delivery,” Chen says.
The Children’s Discovery Institute of Washington University and St. Louis Children’s Hospital, the American Cancer Society, and the National Institutes of Health (NIH) supported the work. Chen recently received a grant from the NIH National Institute of Biomedical Imaging and Bioengineering to work on combining intranasal drug delivery and focused ultrasound with this research.