A low-cost “electronic-nose,” or “e-nose,” uses machine learning to quickly assess beer quality based on its aroma, researchers report.
The device is portable, allowing brewers to use it for rapid and reliable quality assessment on the production line.
It also has the potential to be used to assess other food and beverages at all stages of production and has already been tested to detect smoke taint in wine.
“The e-nose helps take a good deal of subjectivity out of things.”
The development of off-aromas or off-flavors in beers can occur particularly in breweries that don’t use pasteurization.
The partial sterilization that comes through pasteurization isn’t necessary to guarantee the safety of beer as it is a fermented product, and the process can modify aromas. But on the plus side pasteurization can reduce the risk of aroma and flavors going “off.”
Beer flavor boom
With consumers demanding more novelty, brewers are responding by experimenting with different flavor profiles—milky stouts, cherry and honeycomb beers, and even pickle.
In an increasingly innovative environment like this, it’s important to develop methods that can assess aroma both during and after boiling (with the addition of hops) and fermentation.
Brewers can use gas chromatography instruments that analyze the vapors given off by a compound to do this, but it’s costly, time-consuming and involves sending samples away for assessment.
In beer, the aroma alone is a fairly reliable indicator of quality.
This is due to the close link between our senses of smell and taste. But in a small brewery, there may only be one or two people (including the master brewer) who are adequately trained to do this—conducting aroma testing sessions for each batch.
The process can be imperfect and takes time, but it’s also ultimately highly subjective.
New and emerging technologies are able to get around this. Sensors, robotics, and machine learning have been brought together to reduce time spent assessing food quality while providing a less variable test.
“Our study shows that this technology can be used to assess quality in the field. It’s a first for a genuinely portable device,” says lead author Sigfredo Fuentes, associate professor of digital agriculture and food and wine at the Faculty of Veterinary and Agricultural Sciences at the University of Melbourne.
It’s all in the bubbles
The research started with a student project that involved using LEGOs to create a device to measure bubbles in drinks.
The level of bubbling is an important quality of drinks, with consumers preferring medium-sized bubbles in products like sparkling water and in the foam for beer.
“You can determine how much people will like a drink by the quality of the bubbles,” says Fuentes. “Bubbles and foamability and the stability of the bubbles are really important.”
In beer, the froth or foam created by bubbles helps to protect the drink from oxidization that alters the flavor profile—this is why dark stouts like Guinness are often pumped with nitrogen to create a fine foam on top to protect it.
“If you have a flat Guinness, it will oxidize, giving you off-flavors in five minutes,” says Fuentes.
Measuring the bubbles and carbon dioxide led nicely into the next phase of Fuente’s research—adding additional sensors to measure other gases and determine how those bring about different aromas.
Electronic nose vs. the experts
To investigate the effectiveness of the e-nose, the researchers conducted a study of 20 different commercial beer samples, covering a range of styles and fermentation types.
The variety in beer styles helped create a computer program that can detect different volatile compounds, reducing bias in the artificial intelligence software that was developed alongside the e-nose, which Bryce Widdicombe and Ranjith R. Unnithan at the Melbourne School of Engineering built.
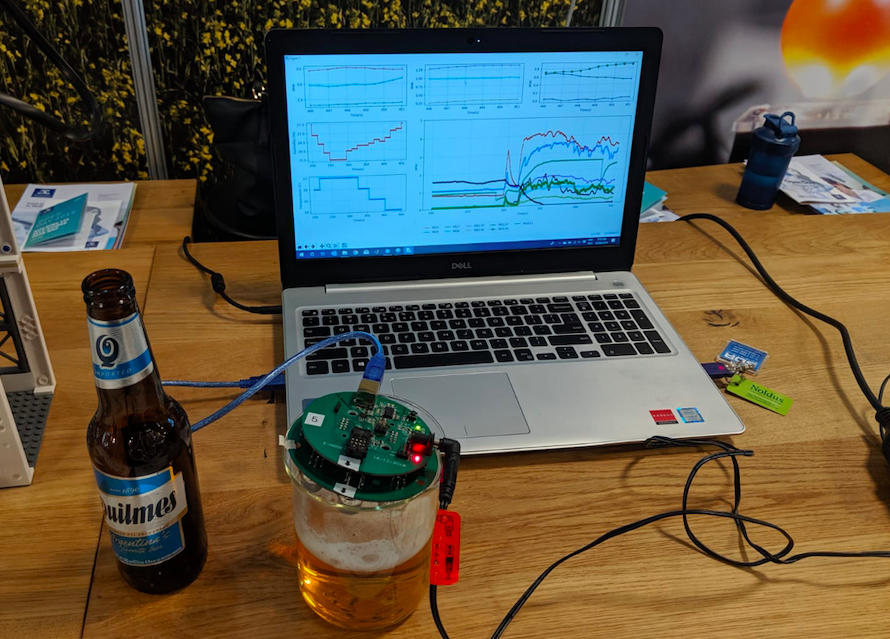
The e-nose—a small circuit board with a diameter of 92 millimeters (about 3.6 inches)—is placed face-down over a sample of beer in a beaker. The sensors can then measure the gases above the beer. This data is measured in real-time.
The researchers measured results against a traditional sensory session conducted with twelve trained panelists.
The panel assessed the intensity of different beer aromas—hops, spicy, floral, fruity, burnt sugar, grains, yeast, nut, and carbonation mouthfeel. The researchers also measured samples using gas chromatography-mass spectroscopy (GC-MS).
One of the objectives is to also use the data from the e-nose to aid in the creation of flavor profiles.
“With the e-nose, you just do a pour, and then you get all the data through the machine learning—you get the whole aroma profile of the beer with 97% accuracy,” says Fuentes.
The e-nose measures the peak area of different gases that are present in beer vapors before fermentation and afterwards.
The sensors on the e-nose are calibrated to measure the presence of gases like carbon dioxide, ethanol, methane, hydrogen, hydrogen sulfide, carbon monoxide, ammonia, and benzene.
There are different gases present as a result of different types of fermentation, like lambic beers. These beers are produced by spontaneous fermentation using wild yeast. This allows for the development of more gases and aromatics than beers from top (like ales) and bottom (like lagers) fermentation that use domesticated yeast varieties.
Looking ahead
The researchers hope to finesse the design further, making it smaller and integrating rechargeable batteries and WiFi to allow brewers greater flexibility, as the current version needs to be connected to power and a computer via USB.
But the potential applications of the device go beyond beer and identifying smoke taint to include building flavor profiles for sparkling wines and non-alcoholic beverages like coffee or tea.
“The e-nose helps take a good deal of subjectivity out of things,” says Fuentes. “Manufacturers will be able to definitively say what is in each flavor profile and put that information on the product labelling.”
But will the e-nose replace a master brewer’s taste? Absolutely not, says Fuentes. “I find that brewers are not against technology. They embrace it.”
The paper appears in Sensors and Actuators B: Chemical.
Source: University of Melbourne



