Researchers have found that a modified ribonucleic acid molecule can “hitchhike” on a plentiful human protein to travel through the blood to a tumor, and then signal that tumor to self-destruct.
Their research gives oncologists a better shot at overcoming the problems of drug resistance, toxicity to patients, and a host of other barriers to consistently achieving successful gene therapy for cancer. It is particularly promising for patients with triple-negative breast cancer, an aggressive type that makes up about 15-20 percent of cases.
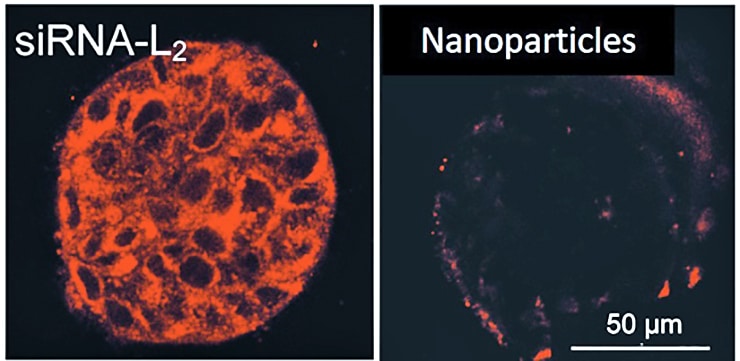
Craig Duvall, associate professor of biomedical engineering at Vanderbilt University, put the effectiveness of a specialized ribonucleic acid hitchhiking on the human protein albumin up against jetPEI nanoparticles, the mostly widely used synthetic carrier for the task of tumor gene silencing.
Ribonucleic acids can control the behavior of cancer cells, but they require a carrier to get them to the target. Duvall’s team made a simple modification to a small-interfering ribonucleic acid molecule, called siRNA-L2, allowing it to rapidly load into an albumin pocket typically reserved to ferry fatty acids around the body.
“Albumin serves almost like a Trojan Horse…”
They found that the siRNA-L2, using albumin as its carrier, has no apparent dose-limiting toxicity, a significant problem for synthetic nanoparticles. That means a higher dose of the anti-cancer drug can be delivered to the tumor without potentially harming the patient.
“Albumin serves almost like a Trojan Horse where it carries it throughout the blood keeps it in the bloodstream for a longer period of time,” says Duvall.
“We used albumin because it’s the highest-concentrated protein in your blood,” he says. “Our molecule, siRNA-L2, binds into the fatty acid pocket of albumin. If we put siRNA directly into the body without a carrier, it’s cleared out by the kidneys in two minutes. If we load siRNA into synthetic nanoparticles to avoid that, then they’re filtered out by the liver.
“Albumin circulates in the body for days, making the siRNA-L2 molecules more available for delivery into tumors,” Duvall says.
Because cancer cells show higher metabolic activity, the albumin that’s carrying siRNA-L2 travels to tumors and gets to work quickly. The molecule’s smaller size allows it to penetrate tumors at a higher rate—with 100 percent of tumor cells testing positive for siRNA-L2 as opposed to only 60 percent when the molecule was carried by jetPEI. Once there, Duvall’s molecule silences a gene crucial to the tumor’s growth and survival.
He says he used the synthetic carrier as a comparison because polymer-based jetPEI represents the gold standard available.
Removing 1 protein may slow cancer cell growth
To make sure their results were translatable to human therapy, the team—in collaboration with Vanderbilt University Medical Center cancer biologist Dana Brantley-Sieders—tested siRNA-L2 in human breast tumor tissue removed from the donor. The Vanderbilt molecule remained more effective, with siRNA-L2 more than three times as present in the tumor than siRNA delivered with synthetic nanoparticles.
Brantley-Sieders says their research has the potential of overcoming the biggest barriers to clinical application of gene-silencing ribonucleic acids.
“What fascinates and excites me most about this approach, in addition to improved tumor penetration, is lack of toxicity at a relatively high dose,” she says.
“We could potentially use our siRNA delivery system to target several genes simultaneously or sequentially. Most cancers are driven by multiple abnormal genes, so targeting one often leads to activation of others as the tumor adapts.”
Targeting these T cells may let immune system kill cancer
The findings appear in the Proceedings of the National Academy of Sciences. The National Institutes of Health and the National Science Foundation provided funding for the research.
Source: Vanderbilt University