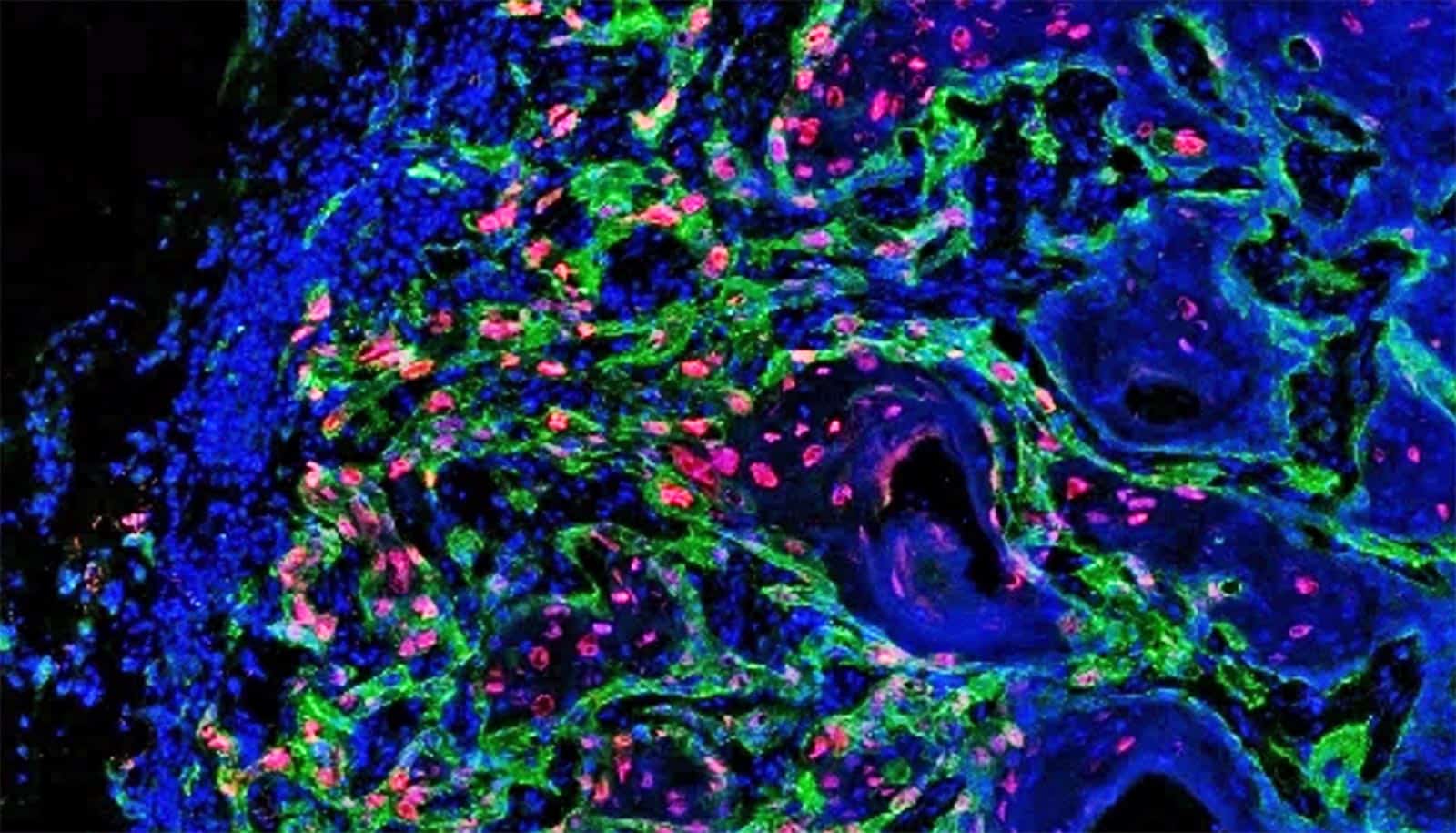
Increasingly dense cell clusters in growing tumors convert blood vessels into fiber-filled channels, research shows.
The findings, from experiments with mice, suggest this makes immune cells less effective.
It was almost ten years ago that researchers first observed that tumors occurring in different cancers—including colorectal cancer, breast cancer, and melanoma—exhibit channels leading from the surface to the inside of the cell cluster. But how these channels form, and what functions they perform, long remained a mystery.
Through a series of experiments, researchers have found possible answers to these questions. There is a great deal of evidence to suggest that these channels, which the researchers have dubbed tumor tracks, were once blood vessels.
These blood vessels start out by supplying the fast-growing cell clusters with glucose and oxygen. But then the vessels undergo a process that strips them of their original function of transporting blood: the vessel walls change and the vessel cavity gradually fills up.
This filler material consists mainly of cells and newly formed protein fibers, which make up what is known as the extracellular matrix. Collagen fibers are found here, as are fibronectin fibers. The latter play a role in growth processes that take place mainly during embryonic development or wound healing. In the journal Matrix Biology, the researchers show that the fibers within the tumor tracks are capable of trapping immune cells.
While this happens, the immune cells stretch out along the channels and stick to the loose fibronectin fibers. “In this elongated form, the immune cells switch from fighting diseases to supporting healing processes,” says Viola Vogel, professor of applied mechanobiology at ETH Zurich. Instead of attacking the tumor cells, they excrete molecules that stimulate growth, thus helping the cancer cells multiply.
It becomes clear that the tension of extracellular matrix fibers plays a key and previously unknown role in tumor development: in healthy tissue, the fibronectin fibers are stretched extremely taut; only in tumor tissue are they slack. In this looser, more relaxed form, surrounded by transformed blood vessel walls, the fibronectin fibers evidently create a recess in which cancer cells can grow undisturbed.
Vogel says that the main focus of cancer research has been on the cells: “The extracellular matrix was frequently overlooked.” That’s why the crosstalk between cells and their environment still remains a mystery. “But if you want to understand what a spider does, you also have to look at its web,” she says.
Vogel therefore also sees the new findings as a reason to expand her research focus and gain a better understanding of the bigger picture. “The better we understand how the microenvironment steers how tumor cells multiply, the likelier it is that we’ll find a way of preventing them from doing so,” she says.
Vogel does, however, caution to translate the implications of the results to humans because they are based on experiments on mice with breast cancer. It remains to be seen whether or not these results can be applied directly to cancers in humans. But there are indeed several parallels, as a group led by Gertraud Orend of the University of Strasbourg recently demonstrated in EMBO Molecular Medicine.
Meanwhile, Vogel’s research group has started collaborating with the Kantonsspital Baden on a follow-up project: one of Vogel’s doctoral students is investigating if tissue samples taken from breast cancer patients also contain traces of converted blood vessels. “We’re curious to discover where we’ll find similarities and where we’ll see differences,” Vogel says.
Source: Ori Schipper for ETH Zurich