A new stimulation device implanted in blood vessels next to the brain’s motor cortex could replace open brain surgery, scientists report.
Surgeons make a small keyhole incision in the neck to place the device, called a Strentrode, which is just 4mm in diameter and is made from a strong but flexible alloy called nitinol.
In 2016 the team, including researchers from the University of Melbourne, Florey Institute of Neuroscience and Mental Health, the Royal Melbourne Hospital and Synchron Australia, demonstrated that the Stentrode can record neural signals in the brain.
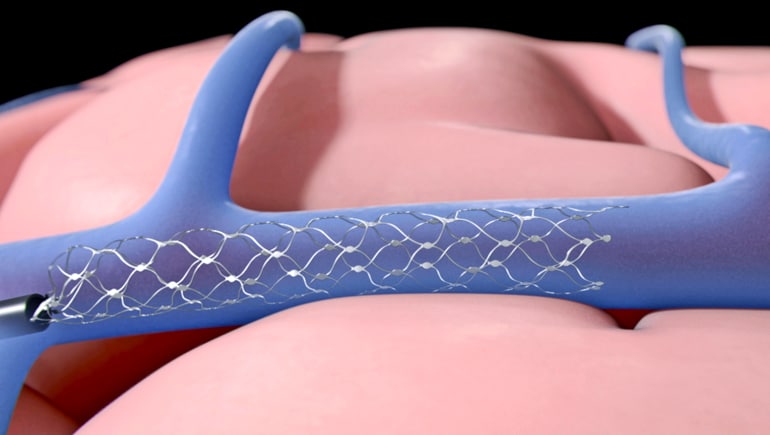
Now, they have shown the same device can not only “listen” to brain signals, but can also “talk back”—delivering currents directly to targeted areas of the brain, a procedure known as “focal brain stimulation.”
Two-way communication
“We were able to not just passively record, but also deliver currents through the device to cause muscle movement,” says Nick Opie from the University of Melbourne’s Vascular Bionics Laboratory and lead researcher of the paper, which appears in Nature Biomedical Engineering.
The proof-of-concept discovery is the first time a device permanently implanted inside a blood vessel stimulated the brain in this way. The range of potential applications is huge, Opie believes.
“There are probably ways the technology could be used that we haven’t even thought of, yet, so we’re really keen to hear from clinicians on their ideas. Some of the obvious applications include offering an alternative to the deep brain stimulation that is currently used to treat Parkinson’s symptoms, and also as a replacement for some drugs in treating certain kinds of epilepsy.”
Perfect touch
In some instances, deep brain stimulation also treats serious mental illnesses like major depression. The team is optimistic that Stentrode could offer these patients a less invasive alternative, too.
Because it can manage two-way communication, essentially acting as a feedback loop within the brain, it may also help people with spinal cord injuries control prosthetic limbs with their brain.
“We can now target both the motor cortex (responsible for planning, control, and execution of movements) and the sensory cortex (which receives feedback about actions) with one device,” Opie says.
“This means we could, for example, help spinal cord patients use a prosthetic arm by commanding it to grab an item, and then providing feedback on that action so they don’t grab it too hard or too soft.”
The device is positioned using a minimally-invasive surgical technique: Surgeons use a microwire and a microcatheter to deploy it at the desired location, guided by “contrast angiography”—an x-ray technique that allows doctors to see inside blood vessels. Once in place, it is operated wirelessly.
Little twitches
“We implanted the device over the motor cortex on the top of the brain, in a small vessel just under the skull,” Opie says.
“The electrodes were positioned along the Stentrode, adjacent to different regions of the brain. By delivering current through these electrodes, we were able to stimulate different brain regions and observe different responses.”
Researchers observed focused muscle movements, including twitches in the neck, lip, and eyes when they stimulated the device.
The next step is to investigate the parameters for stimulation, to discover the lowest possible current the device requires and make it as safe as possible, before progressing to human trials.
“We have a lot of work to do in fine-tuning the parameters,” Opie says. “And these will vary depending on how the device is being used.”
The researchers plan to conduct a clinical trial early next year to help paralyzed people regain movement by enabling them to operate a wheelchair or even an exoskeleton with the device. The ultimate goal is to combine this with the ability to stimulate the brain.
“I am excited to see our technology enhance the quality of someone’s life” Opie says.
Source: University of Melbourne