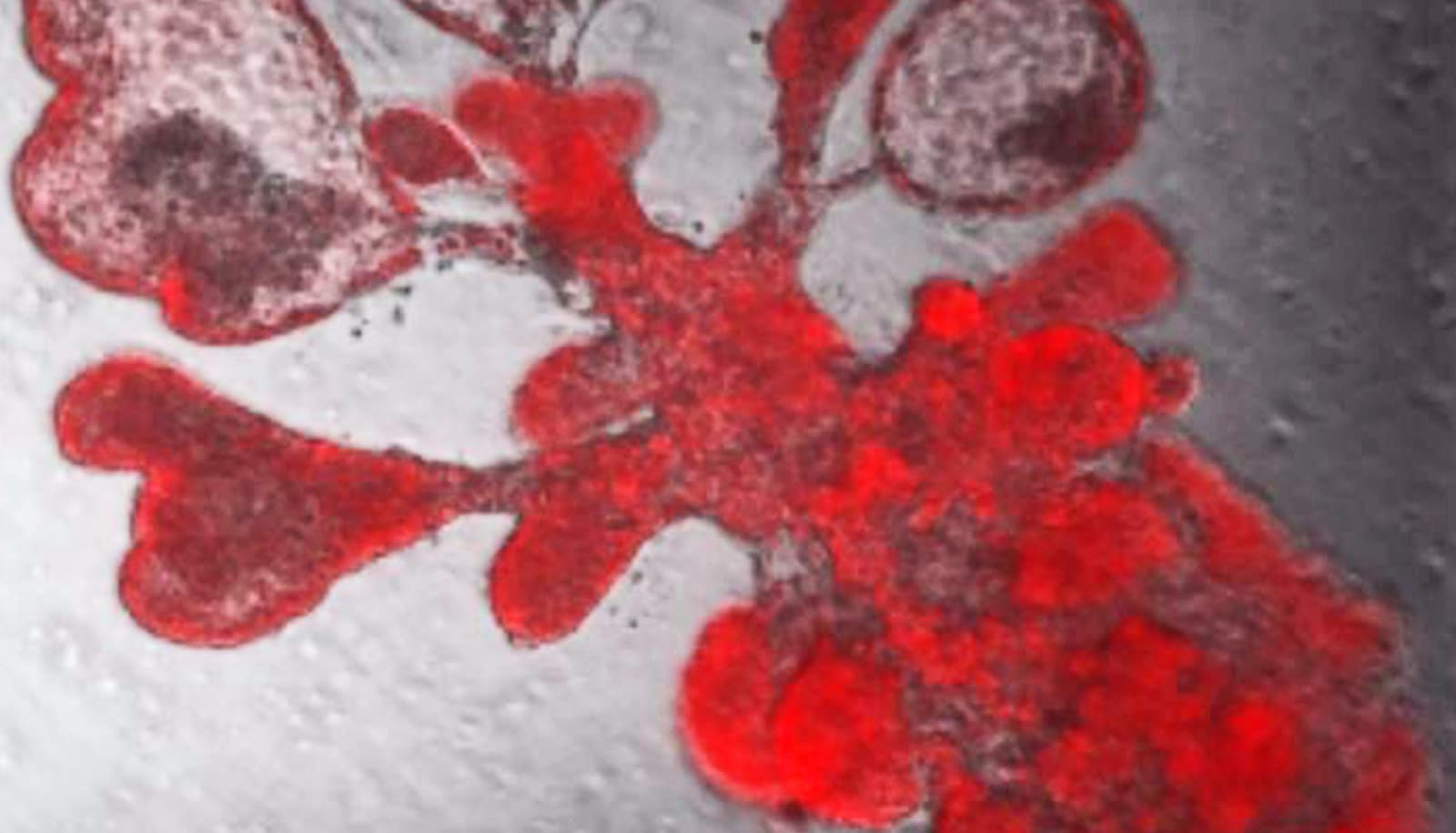
A new lab-grown living lung model mimics the tiny air sacs of the lungs where coronavirus infection and serious lung damage take place.
The advance allows researchers to watch the battle between the SARS-CoV-2 coronavirus and lung cells at the finest molecular scale.
The virus damages the delicate, balloon-like air sacs, known as alveoli, leading to pneumonia and acute respiratory distress, the leading cause of death in COVID-19 patients. But the lack of experimental models that mimic human lung tissues has, until now, hampered scientists in COVID-19 studies.
Purushothama Rao Tata, a cell biologist at Duke University, led a team that developed the model that uses “lung organoids,” also dubbed mini-lungs in a dish. The organoids are grown from alveolar epithelial type-2 cells (AT2s), the stem cells that repair the deepest portions of the lungs where SARS-CoV-2 attacks.
Earlier research had shown that just one AT2 cell, isolated into tiny dishes, could multiply to produce millions of cells that assemble themselves into balloon-like organoids that look just like alveoli. However, the “soup” in which researchers grew the cells contained complex ingredients such as serum from cows that is not completely defined.
100% human mini-lungs
For the new study, published in Cell Stem Cell, Tata’s group took on the big challenge of predicting and testing many combinations of chemically pure factors that would do the job just as well.
The result is a purely human organoid without any helper cells. Mini-lungs grown in tiny wells will enable high throughput science, in which hundreds of experiments can be run simultaneously to screen for new drug candidates or to identify self-defense chemicals produced by lung cells in response to infection.
“This is a versatile model system that allows us to study not only SARS-CoV-2, but any respiratory virus that targets these cells, including influenza,” Tata says.
In using mini-lungs to study SARS-CoV-2 infection, the researchers had to take precautions to safely handle the deadly virus. The researchers utilized state-of-the art biosafety level 3 facilities to infect the lung organoids. The researchers watched the gene activity and chemical signals that are produced by the lung cells after infection.
“This is a major breakthrough for the field because we were using cells that didn’t have purified cultures,” says coauthor Ralph Baric, a professor of epidemiology, microbiology, and immunology at the University of North Carolina at Chapel Hill.
The mini-lungs are 100% human with no supporting cells that could confuse findings. “This is incredibly elegant work to figure out how to purify and grow AT2 cells in culture in pure form,” Baric says.
New weapon in COVID fight
Baric’s lab is capable of changing any nucleotide of the COVID-19 virus’s genetic code at will, so it produced a glowing version that would reveal where it went in the mini-lungs, confirming that it did indeed home in on the crucial ACE2 cell surface receptor, leading to infection.
When infected with the virus, the organoids launched an inflammatory response mediated by interferons. The researchers have also witnessed the cytokine storm of immune molecules the lungs launch in response to the virus.
“It was thought cytokine storm happened due to the large influx of immune cells, but we can see it also happens in the lung stem cells themselves,” Tata says.
Tata’s lab found the cells produced interferons and experienced self-destructive cell death, just as samples from COVID-19 patients have shown. The signal for cell suicide was sometimes triggered in uninfected neighboring lung cells as well, as the cells struggled to get ahead of the virus.
The researchers also compared the gene activity patterns between the mini-lungs and samples from six severe COVID-19 patients and found they agreed with “striking similarity.”
“We’ve only been able to see this from autopsies until now,” Tata says. “Now we have a way to figure out how to energize the cells to fight against this deadly virus.”
Cell-based therapies
In another series of experiments, mini-lungs treated with low doses of interferons before infection were able to slow viral copying. But suppressing interferon activity before infection led to increased viral replication.
Tata says his lab was working on growing the mini lungs in mid-2019 and had achieved a working model just as the coronavirus pandemic emerged.
He says his group will be working with both academic and industry partners to use these cells for cell-based therapies and eventually to try to grow a complete lung for transplantation.
Baric says his lab will probably use the mini-lungs to better understand a new strain of SARS-CoV-2 called D614G that has become the dominant version of the virus. This strain, which emerged in Italy, has a spike protein that is apparently more efficient at recognizing the ACE2 receptor on lung cells, making it even more infectious.
The Chan Zuckerberg Foundation, the US National Institutes of Health, Duke University, and United Therapeutics Corporation funded the work.
Source: Duke University