Artisanal makers of mezcal use bubbles to tell when the drink has the right alcohol level. New research reveals the physics behind this skill.
Makers of mezcal squirt some into a small container and look for little bubbles, known as pearls. If the alcohol content is too high or too low, the bubbles burst quickly. But if they linger for 30 seconds or so, the alcohol level is perfect and the mezcal is ready to drink.
Using laboratory experiments and computer models, fluid dynamics researchers show that a phenomenon known as the Marangoni effect helps mezcal bubbles linger a little longer when the alcohol content is around the sweet spot of 50%. In addition to showing the scientific underpinnings of something artisans have known for centuries, the researchers say the findings reveal new fundamental details about the lifetimes of bubbles on liquid surfaces.
The study appears in the journal Scientific Reports.
When Roberto Zenit, a professor in Brown University’s School of Engineering and the study’s senior author, first heard about the bubble trick, he says he was instantly intrigued.
“One of my main research interests is bubbles and how they behave,” Zenit says. “So when one of my students told me that bubbles were important in making mezcal, which is a drink that I really enjoy with my friends, it was impossible for me not to investigate how it works.”
Bubble lifetimes and alcohol level
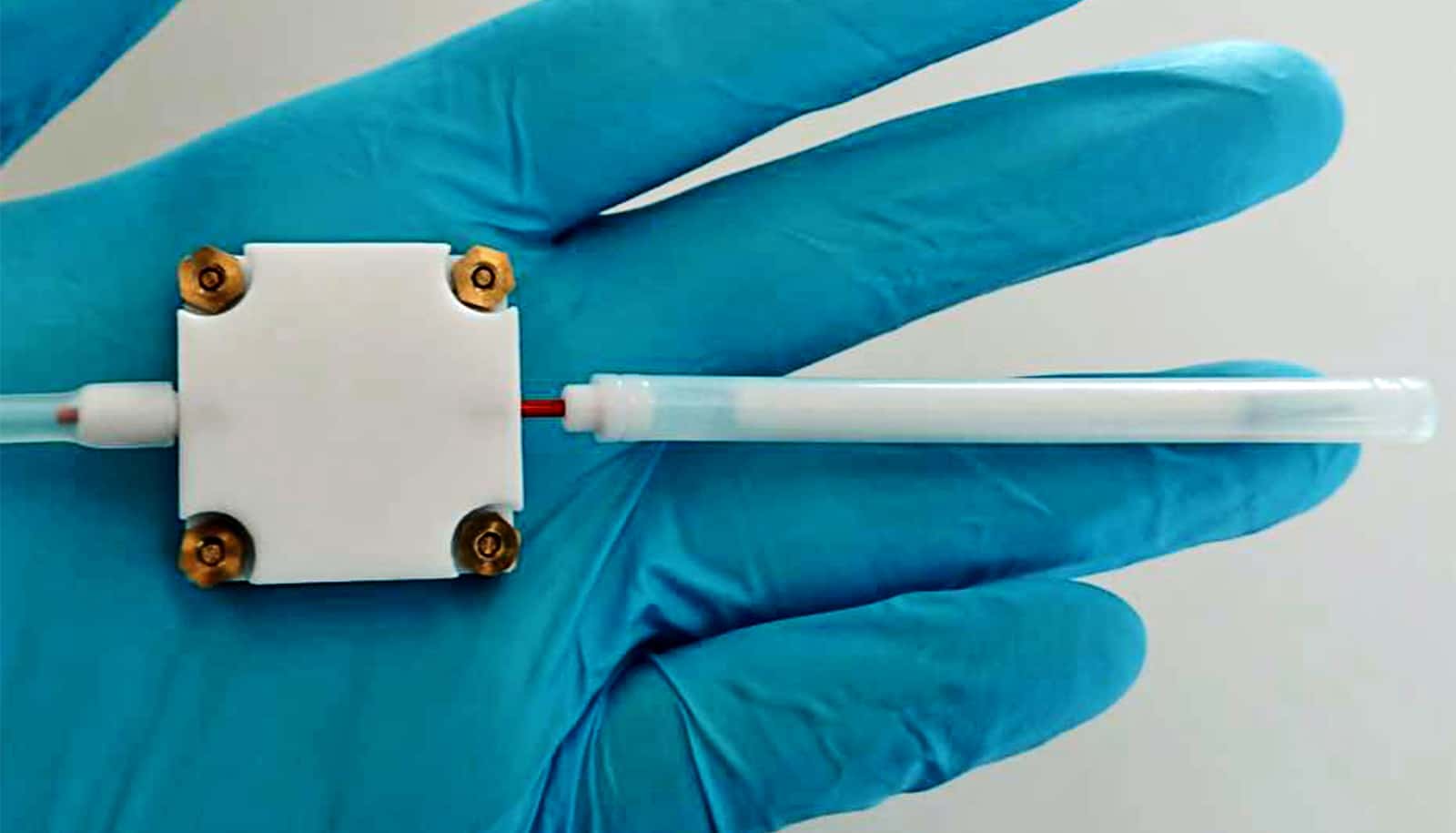
The researchers started by doing experiments to see how changing the alcohol level of mezcal changed bubble lifetimes. They watered down some samples of mezcal and added pure ethyl alcohol to others. They then reproduced the squirting trick in the lab while carefully timing the bubbles. They found that, sure enough, alcohol level dramatically affected bubble lifetimes. In unaltered samples, bubbles lasted from 10 to 30 seconds. In both the fortified and watered-down samples, the bubbles burst instantly.
Having shown that bubbles really can be a gauge of alcohol content, the next step was to figure out why.

To do that, the researchers started by simplifying the fluid—performing experiments with mixtures of just pure water and alcohol. Those experiments showed that, as with mezcal, bubbles tended to last longer when the mixture was near 50% water and 50% alcohol. The researchers determined that the extra bubble life was due largely to viscosity. Bubbles tend to last longer in more viscous fluids, and the viscosity of alcohol-water mixtures peaks right around 50%.
However, the bubbles in the 50-50 water and alcohol mixtures still didn’t last as long as those in mezcal. Zenit and his students realized there must be something about mezcal that amplifies the viscosity effect. To figure out what it was, they used high-speed video cameras to carefully watch the bubbles through their lifetimes.
The video revealed something surprising, Zenit says. It showed an upward convection of liquid from the surface of mezcal into the bubble membranes.
“Normally, gravity is causing the liquid in a bubble film to drain away, which eventually causes the bubble to burst,” Zenit says. “But in the mezcal bubbles, there’s this upward convection that’s replenishing the fluid and extending the life of the bubble.”
Marangoni convection in mezcal
With the help of some computer modeling, the researchers determined that a phenomenon known as the Marangoni convection was responsible for this upward motion. The Marangoni effect occurs when fluids flow between areas of differing surface tension, which is the attractive force between molecules that forms a film surface of a fluid. Mezcal contains a variety of chemicals that act as surfactants—molecules that change the surface tension. As a result, bubbles that form on the surface of mezcal tend to have higher surface tension than the surfactant-filled fluid below. That differing surface tension draws fluid up into the bubble, increasing its lifespan.
By amplifying the existing tendency for longer-lasting bubbles in 50% alcohol mixtures, the surfactant-driven Marangoni effect makes bubbles a reliable gauge of alcohol content in mezcal.
Zenit, who hails from Mexico, says it was gratifying to shed new light on this artisanal technique.
“It’s fun to work on something that has both scientific value and cultural value that’s part of my background,” he says. “These artisans are experts in what they do. It’s great to be able to corroborate what they already know and to demonstrate that it has scientific value beyond just mezcal making.”
The insights generated from the work could be useful in a variety of industrial processes that involve bubbles, the researchers say. It could also be useful in environmental research.
“For example,” the researchers write, “the lifetime of surface bubbles could be used as a diagnostic tool to infer the presence of surfactants in a liquid: If the lifetime is larger than that expected of a pure/clean liquid, then the liquid is most likely contaminated.”
Source: Brown University