Modulating gut bacteria could enhance the therapeutic value of a drug, research with the Alzheimer’s medication tacrine suggests.
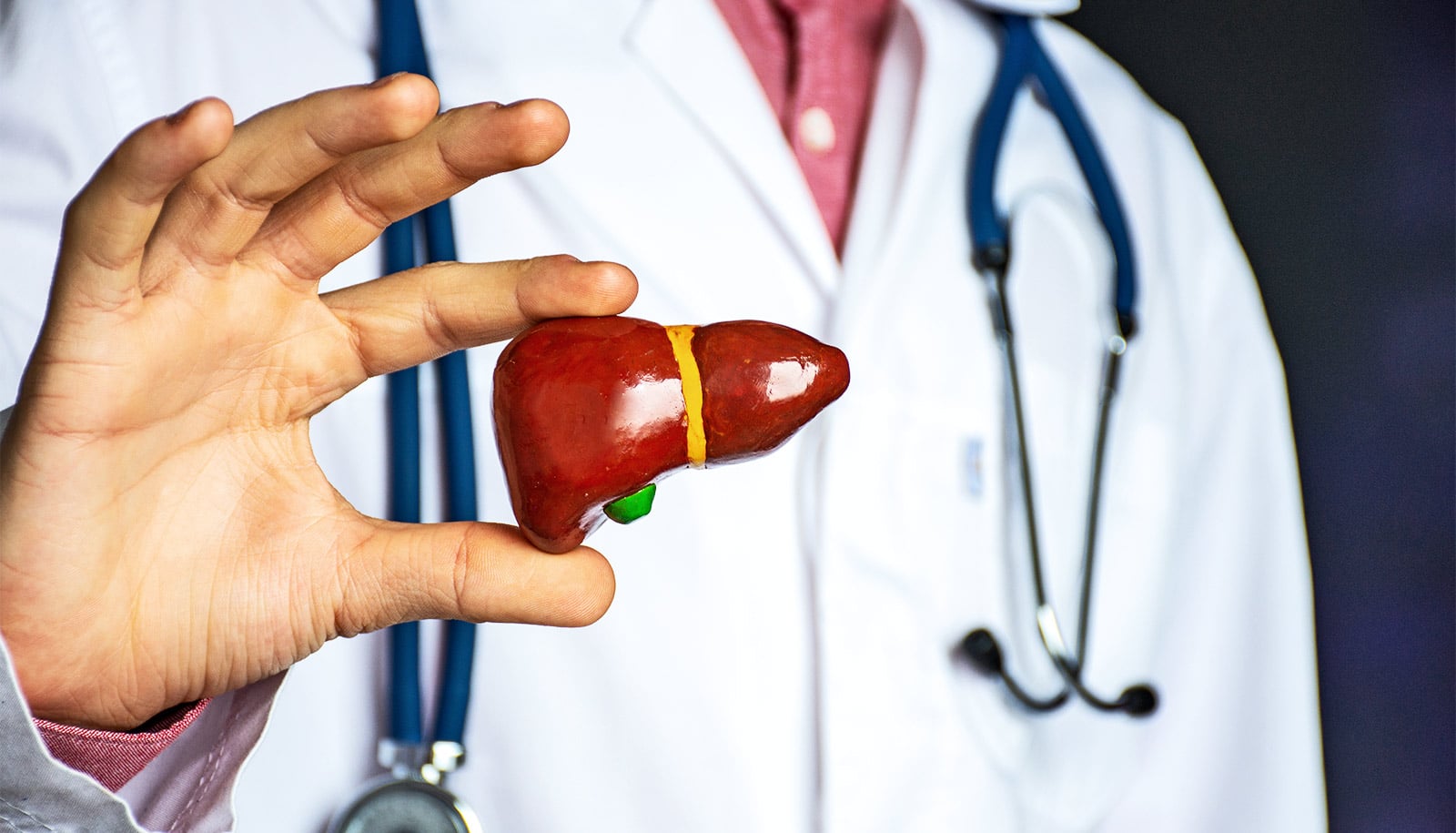
Enzymes in the liver break down pharmaceutical drugs into their metabolites, which the body then excretes in feces and urine. Drugs can also undergo chemical reactions mediated by bacteria within the gut.
“To date, the influence of microbes on the fate of drugs is only established for a small fraction of marketed medicines.”
In a new study, a team of pharmaceutical scientists from National University of Singapore found that gut bacteria may selectively modify the breaking down of tacrine. This causes certain patients to suffer from drug-induced liver injury (DILI), in which there are unexpected side effects from consuming a drug, causing liver damage.
The discovery and understanding of this process could potentially lead to the development of new ways to improve drug treatment outcomes and reduce drug-associated side effects.
“To date, the influence of microbes on the fate of drugs is only established for a small fraction of marketed medicines. The contribution of gut microbiota towards variation in the response of individuals to a given drug remains poorly understood. This difference in the response to drug therapy is clinically important and poses a major challenge to both drug development and patient management,” says study leader Eric Chan from the department of pharmacy.
The research team found that rats suffering from DILI experienced a process termed “enhanced enterohepatic recycling,” where the excreted metabolite of the drug in bile is reconverted back to the parent drug by gut bacteria and reabsorbed into the body from within the gut.
As reported in the journal Hepatology, the researchers found that these DILI rats had a greater amount of a specific bacteria that can enable this drug reconversion process. Deactivating these bacteria resulted in less injury to the liver.
Could compound in aged cheese spare our livers?
“Our study establishes the gut microbial influences which cause unintended liver damage when using the drug tacrine, providing crucial insights for the development of personalized medicine initiatives,” explains first author Yip Lian Yee.
“Our study confirms for the first time that the liver-gut microbial community has an influence on unintended liver damage caused by tacrine in rodent models,” adds Chan.
“While this study is focused on tacrine, our findings hold pertinent implications for other therapeutic drugs with side effects, particularly those that undergo extensive enterohepatic recycling, demonstrate large variations between individuals and exhibit narrow therapeutic indices.”
The research took place in collaboration with scientists from GlaxoSmithKline, the NUS departments of microbiology and surgery, and the Genome Institute of Singapore (A*STAR).
Source: National University of Singapore