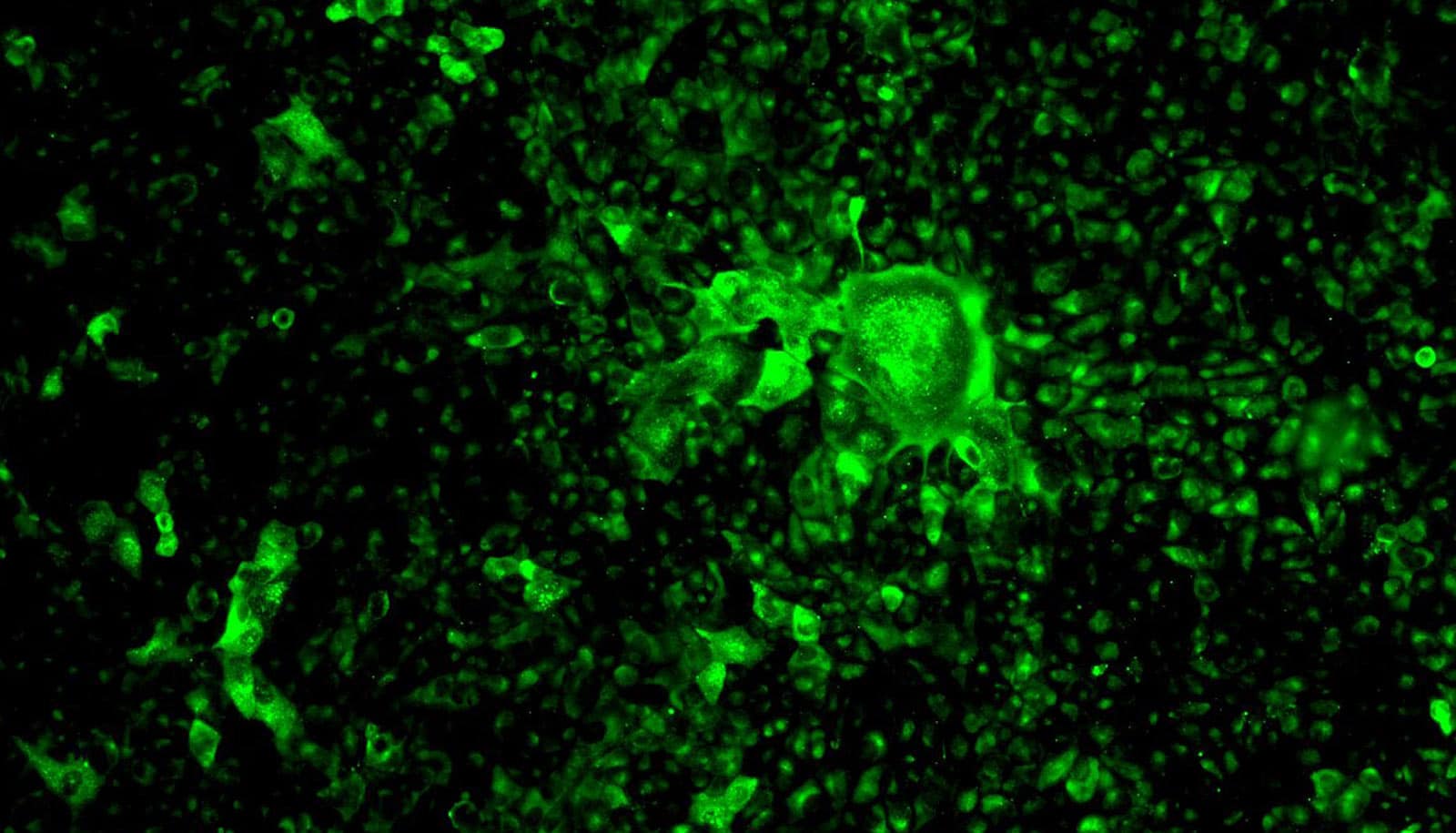
Scientists say they’ve found a way to light up the SARS-CoV-2 virus using glowing antibodies, which makes it possible to detect the virus as it infects laboratory cell cultures.
“We can now see the enemy—it’s like switching on the lights in a dark room,” says Robert Davey, microbiologist at Boston University’s National Emerging Infectious Diseases Laboratories (NEIDL).
It’s the first major step forward in his team’s SARS-CoV-2 research that began on March 19, 2020. They and other teams at NEIDL—including one group examining SARS-CoV-2 and the immune response it inflames in animals—are the only scientists in New England working with live copies of the novel coronavirus.
Davey’s team specializes in pitting thousands upon thousands of drugs—small molecules made of different chemical concoctions—against lab cultures of cells infected with contagions, allowing them to rapidly detect which drugs are most effective at halting or reducing infection. Now that they’ve effectively got eyes on the SARS-CoV-2 virus’s whereabouts inside the cells it infects, the team is ready to screen upwards of 20,000 drug compounds to test their efficacy in halting or reducing COVID-19 infections.
The number of compounds the team expects to test has more than doubled in the two weeks since news of their research gained national attention. “It seems like we have an almost infinite number of compounds to test,” Davey says. “I’ve had an avalanche of email and telephone calls from biotech and pharma companies.”
The antibodies that will allow Davey’s team to see SARS-CoV-2 are made of the same parts and proteins as the antibodies the human immune system produces in response to COVID-19 infection. Like your own antibodies, they are designed to zero in on SARS-CoV-2 and interlock with a perfectly fitted keyhole, made of proteins, on the surface of the virus. In humans, antibodies glom onto viruses to flag them for disposal by killer immune cells, which patrol our bodies and destroy any foreign materials identified by antibodies.
Davey’s team is using antibodies for a different purpose.
“You use one antibody that detects the presence of the virus—then on top of that you link another one that fluoresces under blue light,” Davey says. “You end up with a little stack of antibodies attached to SARS-CoV-2, and the last antibody on the tower lights up the virus’s location.”
But to create that perfect interlocking stack of antibodies, Davey’s team had to test a number of them. “We have very particular requirements—we need these antibodies to stick together really tight and give us a good signal [via fluorescence],” Davey says. “They act like Velcro or keys into locks; you want the key to fit really well into the lock.”
When the key holds into its lock really well, Davey says, that allows them to speed up their drug testing process, which requires physical agitation of the virus and infected cells. With the flood of new compounds that Davey’s team has been sent to screen, the ability to work quickly will make all the difference. To get ready, he called up lab equipment provider BioTek Instruments to order another cell-imaging machine. Instead, the company donated one without hesitation. “They said, ‘OK, we’re sending one.’ They didn’t even ask for money,” Davey says. “It takes a village—we’re all on the same team.”
Typically, Davey’s team handles some of the world’s most lethal diseases, like Ebola or Marburg fevers, inside a Biosafety Level 4 (BSL-4) laboratory, which has the highest possible level of biosafety containment used for infectious agents that pose especially high risk to humans. BSL-4 is a full step of containment above the required BSL-3 that’s needed for working with live copies of SARS-CoV-2.
But the personal protection equipment that researchers must wear for BSL-3 work is the same gear that hospital clinicians are direly short of right now. So, at the NEIDL, one of the few research facilities in the US to have both BSL-3 and BSL-4 laboratories, Davey and the members of his team will handle the SARS-CoV-2 virus while wearing full, airtight biocontainment suits, each with its own oxygen supply, taking the virus up to the BSL-4 level of containment.
Source: Boston University