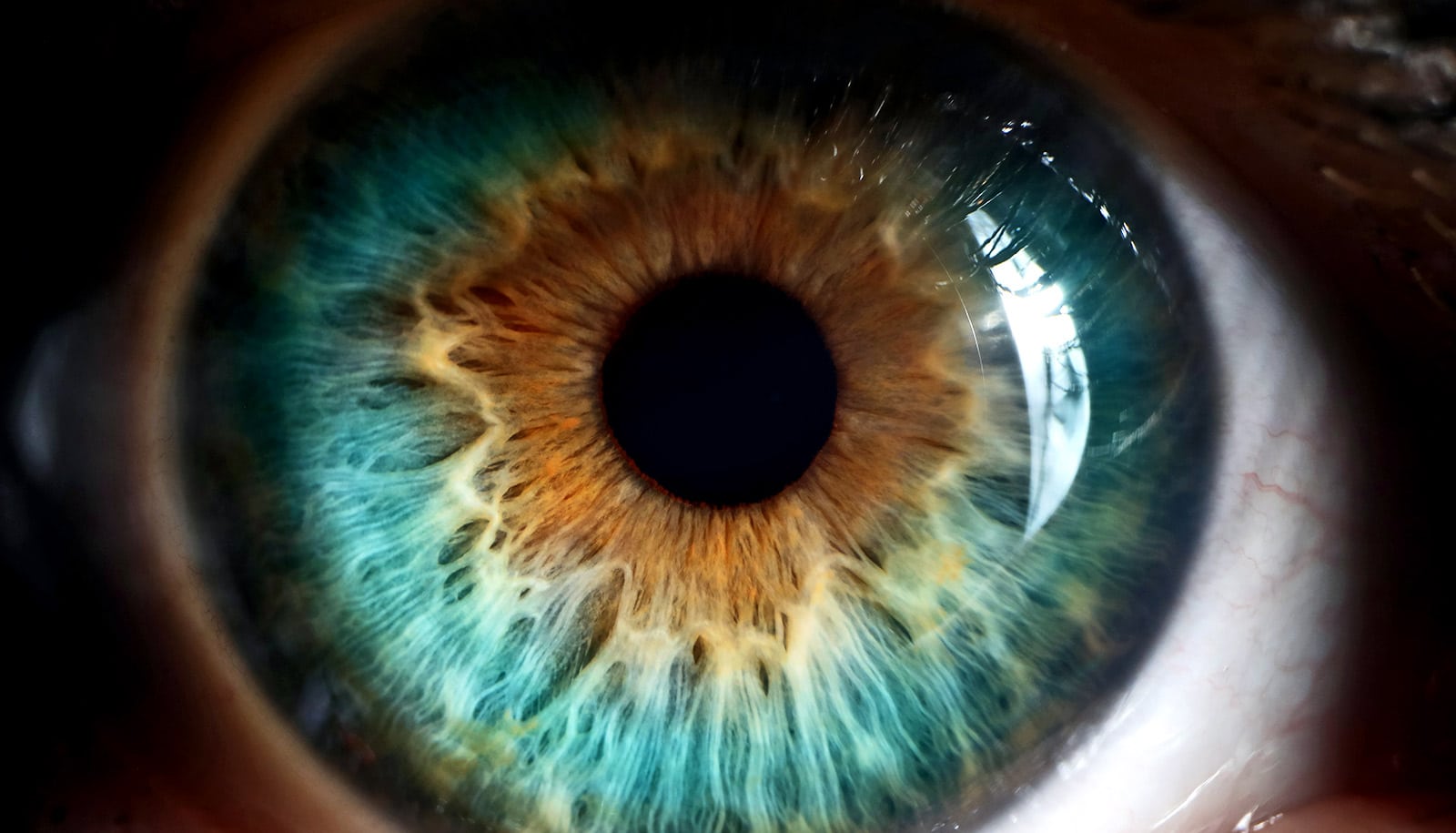
A biomarker could be a way to monitor how fast glaucoma is progressing, as well as the effectiveness of treatment.
“There hasn’t been a reliable way to predict which patients with glaucoma have a high risk of rapid vision loss,” says principal investigator Rajendra S. Apte, professor of ophthalmology and visual sciences at Washington University in St. Louis.
“But we’ve identified a biomarker that seems to correlate with disease severity in patients, and what that marker is measuring is stress to the cells rather than cell death. Other glaucoma tests are measuring cell death, which is not reversible, but if we can identify when cells are under stress, then there’s the potential to save those cells to preserve vision.”
Glaucoma is the second leading cause of blindness in the world, affecting more than 60 million people. The disease often begins silently, with peripheral vision loss that occurs so gradually that it can go unnoticed. Over time, central vision becomes affected, which can mean substantial damage already has occurred before any aggressive therapy begins.
Many patients start receiving treatment when their doctors discover they have elevated pressure in the eye. Those treatments, such as eye drops, are aimed at lowering pressure in the eye, but such therapies may not always protect ganglion cells in the retina, which are the cells destroyed in glaucoma, leading to vision loss.
All current treatments for glaucoma are aimed at lowering pressure in the eye to reduce ganglion cell loss and not necessarily at directly preserving ganglion cells, says Apte, who is also a professor of developmental biology, of medicine, and of neuroscience.
New way of imaging eyes could spot glaucoma sooner
Glaucoma specialists attempt to track vision loss caused by ganglion cell death with visual field testing where a patient pushes a button when they see a blinking light.
As vision is lost, patients see fewer lights blinking in the periphery of the visual field, but such testing is not always completely reliable, says Norimitsu Ban, an ophthalmologist and postdoctoral research associate in Apte’s laboratory. Ban is first author of the study in JCI Insight.
Some older people don’t do as well on the visual field test for reasons that may not be related to what’s going on in their eyes, Ban says, so finding a marker of cell damage in the eye would be a much more reliable way to track the progression of glaucoma.
“We were lucky to be able to identify a gene and are very excited that the same gene seems to be a marker of stress to ganglion cells in the retinas of mice, rats and humans,” he says.
Studying mouse models of glaucoma, the researchers identified a molecule in the eye called growth differentiation factor 15 (GDF15), noting that the levels of the molecule increased as the animals aged and developed optic nerve damage.
When they repeated the experiments in rats, they replicated their results. Further, in patients undergoing eye surgery to treat glaucoma, cataracts, and other issues, the researchers found that those with glaucoma also had elevated GDF15 in the fluid of their eyes.
“That was exciting because comparing the fluid from patients without glaucoma to those with glaucoma, the GDF15 biomarker was significantly elevated in the glaucoma patients,’ Apte says. “We also found that higher levels of the molecule were associated with worse functional outcomes, so this biomarker seems to correlate with disease severity.’
The researches don’t believe that the molecule causes cells in the retina to die; rather, it’s a marker of stress in retinal cells. “It seems to be a harbinger of future cell death rather than a molecule that’s actually damaging the cells,” Apte says.
A potential limitation of the study is that the fluid samples were taken from the eyes of patients only once, so it was not possible to monitor levels of GDF15 over time. In future studies, it will be important to measure the biomarker at several time points to determine whether levels of the biomarker increase as the disease progresses.
Apte would like to know whether GDF15 levels eventually decline in those who have significant vision loss from glaucoma. In theory, when most of the ganglion cells in the retina already have died, fewer cells would be under stress, and that could mean lower levels.
“So we are interested in doing a prospective study and sampling fluid from the eye over several months or years to correlate glaucoma progression with levels of this marker”‘ he says. “We’d also like to learn whether levels of GDF15 change after treatment, a particularly important question as we try to develop therapies that preserve vision more effectively in these patients.”
The National Eye Institute, the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Neurological Disorders and Stroke, and the National Institute of General Medical Sciences, of the National Institutes of Health funded the work.
The Schulak Family Gift Fund for Retinal Research, the Jeffrey Fort Innovation Fund, the Kuzma Family Gift Fund, the Central Society for Clinical and Translational Research, a Research to Prevent Blindness Physician Scientist Award, the Washington University Institute of Clinical and Translational Sciences, the American Federation for Aging Research, the Vitreoretinal Surgery Foundation and an unrestricted grant from Research to Prevent Blindness Inc. provided additional funding.
Source: Washington University in St. Louis