New research could open new paths for drug treatment for a widespread parasite.
It infects nearly one-third of the global population, yet its microscopic size makes the parasite difficult for scientists to study.
That parasite is Toxoplasma gondii, a widespread organism that infects humans and animals.
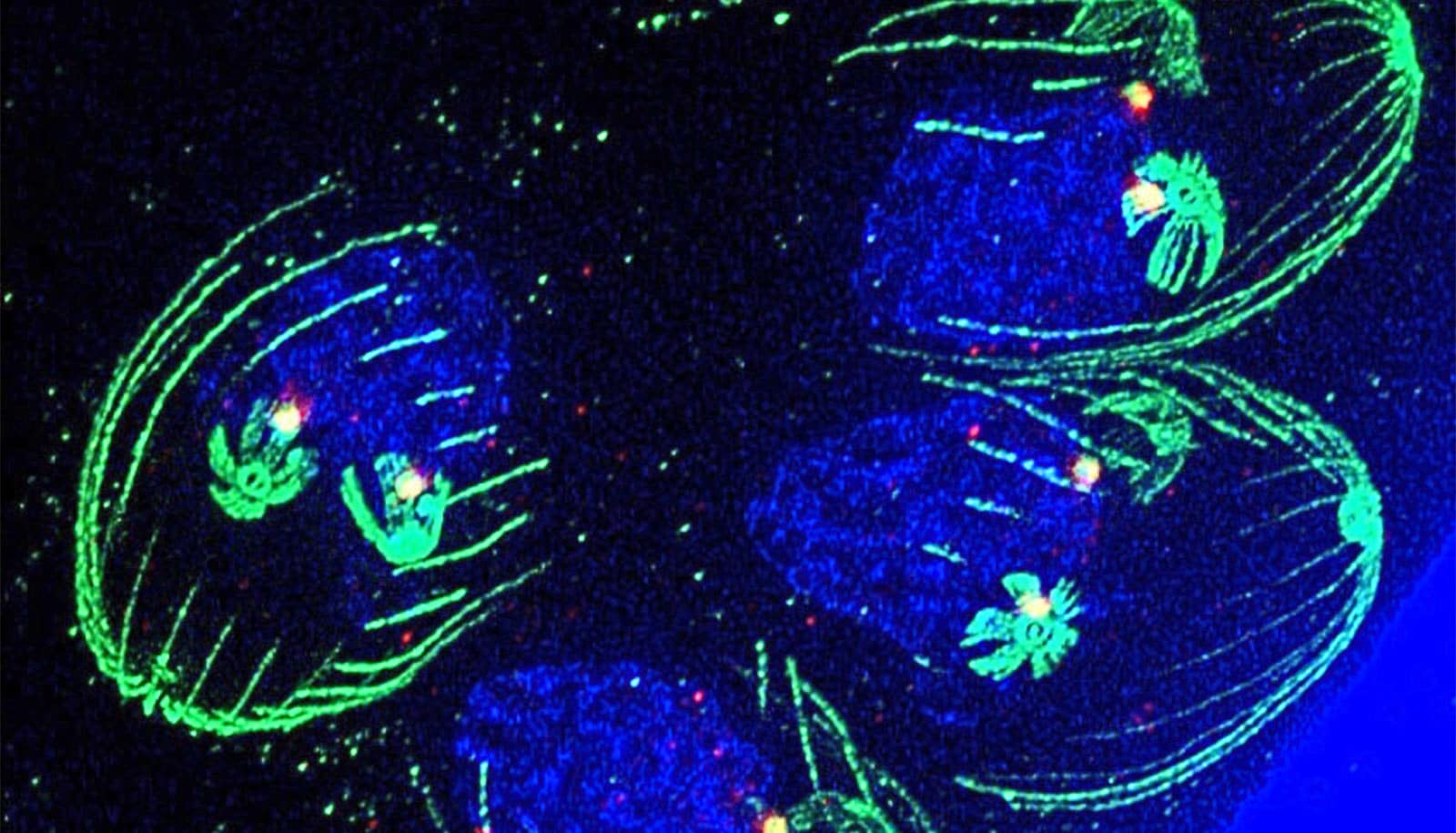
To better understand how it functions, infectious disease researchers at the USF Health Morsani College of Medicine adapted a fluorescent imaging system typically used to study human cells to observe the parasite’s growth in real time—paving the way for future treatments.
Toxoplasma is commonly spread through uncooked meat and contaminated produce. When the parasite enters the human body, it causes toxoplasmosis, an infection that is often mild, but can be serious for pregnant women and people with weakened immune systems. It can be treated if caught within the first two weeks of exposure.
“Though the parasite can be repressed in the acute stage, it requires drugs that can be toxic if taken long term,” Suvorova says.
“If you can’t catch toxoplasmosis during this time, the parasite turns chronic. In this stage, it hides from the immune system and forms cysts in the brain, for which there are currently no cures.”
Developing improved treatments has been challenging in part because of the parasite’s unusual cell cycle. A typical cell cycle begins with the cell growing larger before making a complete copy of its DNA. Once everything is prepared for division, the cell splits into two identical parts.
“Toxoplasma doesn’t follow this standard pattern,” says Mrinalini Batra, a research scientist in the Suvorova Lab.
“Scientists knew it had to go through similar stages because it reproduces, but they didn’t know how those stages were arranged or whether they even existed in the same way as they do in human cells. That made it hard to understand how this parasite grows and spreads.”
The goal of the researchers wasn’t just basic curiosity, but part of a bigger effort to eventually stop the parasite from multiplying. To do that, the team needed to map out how its cell cycle works and in what order.
To tailor their fluorescent imaging model for Toxoplasma, the team first had to identify proteins that appear in specific growth stages of the parasite. These proteins also needed to be located in structures that are large enough to visualize, such as the cell’s nucleus, and required fluorescent colors bright enough to stand out in such a tiny organism under a microscope.
Because Toxoplasma lacks many of the common proteins found in human cells, the process required extensive trial and error. The team tested different parts of the parasite using red and green fluorescent tags, but many markers either failed to glow brightly enough or did not appear in sufficient amounts to be useful.
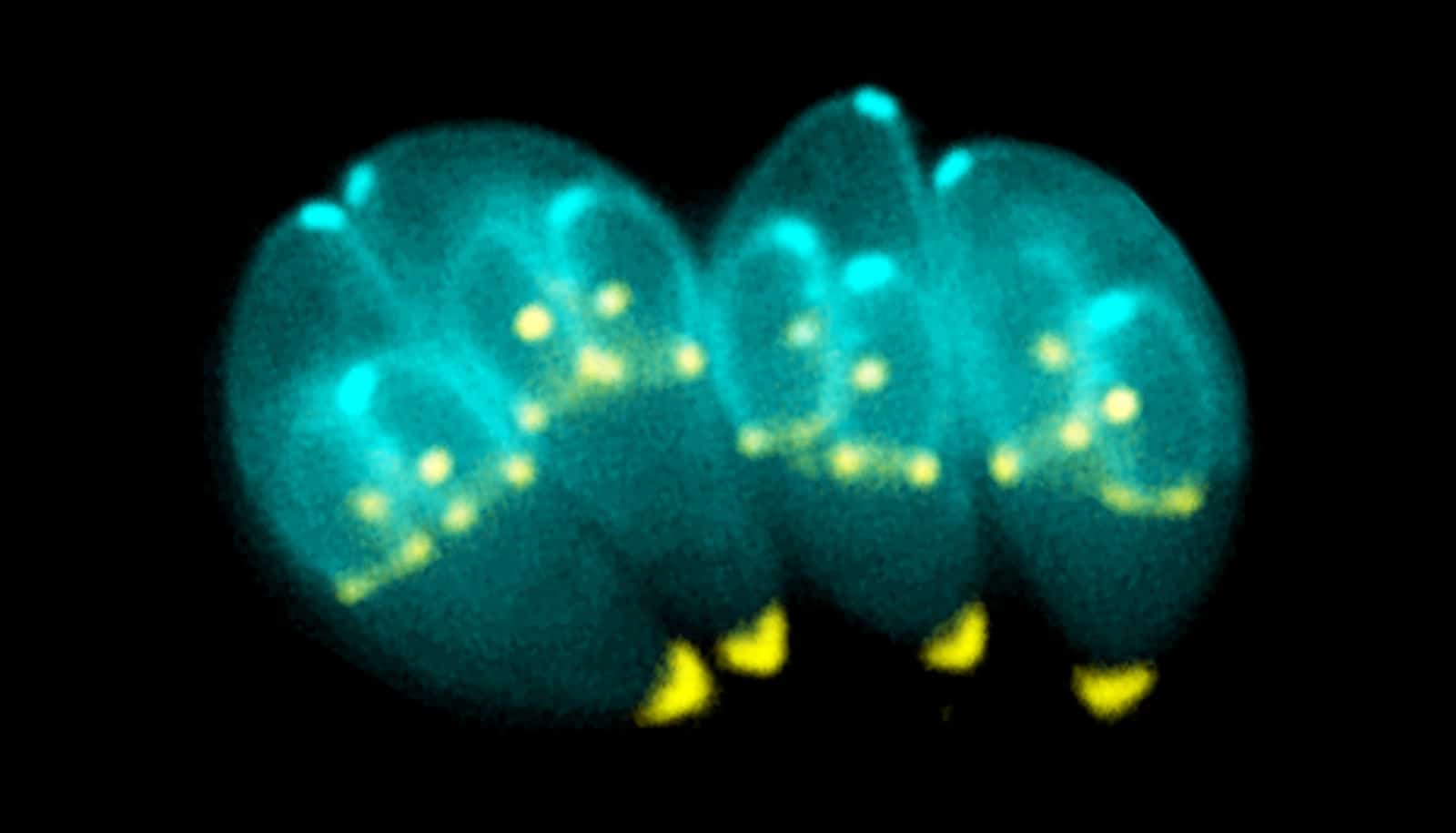
As the team tested multiple combinations, they ultimately identified a protein called PCNA1. This protein is located in the parasite’s nucleus and naturally shifts as the organism progresses through its growth cycle.
“When we attached two copies of a bright neon green tag to this protein, the signal became strong and clear,” Batra says. “This allowed us to determine the parasite’s stage simply by watching how the glowing protein behaved in the cell cycle. For the first time, researchers were able to clearly map Toxoplasma’s cell cycle.”
The discovery shows how the parastate moves normally through the first part of its cell cycle, but the rest of its growth stages overlap instead of occurring sequentially.
“These latter stages are similar to a fork’s structure,” Suvorova says. “Toxoplasma’s cell cycle begins with one straight handle and then several prongs that branch off, allowing as many as three cell cycle phases to occur simultaneously. This unusual pattern helps the parasite multiply rapidly and evade the host’s immune system before forming cysts in the brain.”
Now that Toxoplasma’s cell cycle has been mapped through fluorescence, the team is working to identify weak points in the parasite that could prevent it from multiplying. The team is also testing how different drugs affect specific stages of the cycle in hopes of developing safer and more effective treatments.
The research appears in mBio.
Source: University of South Florida