Using a new live-imaging technique, researchers directly detected short-lived leaks in tissues that protect us from bacteria, viruses, and other disease-causing invaders.
Epithelial tissues are sheets of cells that coat our organs, creating wall-like barriers for protection. And when potentially harmful gaps between these cells emerge, a molecular switch gets flipped to call the repair crew and fix the leaks.
The researchers’ new microscopy barrier assay also allowed them to discover that the repair mechanism involves local activation of a protein called Rho.

The new assay could help provide insights into the mechanisms of diseases that target the epithelial barrier—ailments that microbes and allergens cause, as well as various inflammatory states, immune disorders, diabetes, and even cancers. And the assay could potentially help screen drugs to treat those afflictions, according to the researchers.
“An important unanswered question about epithelial tissues is: How are the junctions between cells able to maintain the biological barrier function even as cells change shape?” says senior author Ann Miller, an associate professor in the molecular, cellular, and developmental biology department at the University of Michigan.
In their study, the researchers used epithelial cells in live frog embryos, which have cell-cell junctions similar in structure and protein composition to those in human epithelial tissues.
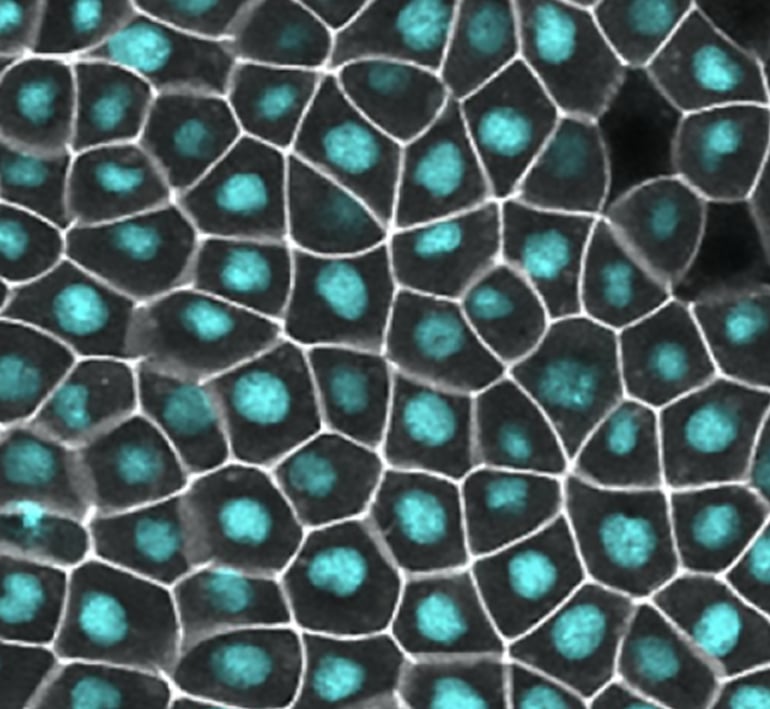
During embryonic development, many epithelial cells work together to bend and fold tissues. Using their new microscopy assay—which is known as ZnUMBA for Zinc-based Ultrasensitive Microscopic Barrier Assay—the researchers studied what happens at the cellular level when epithelial cell-cell junctions are stretched.
They showed that leaks in barrier function happened when cell-cell junctions elongate. But the leaks are short-lived, suggesting there is an active repair mechanism.
On further investigation, the researchers discovered that the repair mechanism involves local activation of the protein Rho, in a sudden burst of activity they dubbed a Rho flare. Rho then activates proteins that contract the junction, repairing it.
“We discovered that cells are normally very proactive when it comes to maintaining the barrier,” says first author Rachel Stephenson, a research scientist in Miller’s lab who carried out the project for her doctoral dissertation.
“This repair mechanism happens quickly and is carried out very locally, affecting only a small part of the cell junction, rather than multiple cells or the whole tissue.
“We think that this proactive approach is what gives our cells the flexibility to move and change shape without compromising the barrier function of the tissue. Diseases involving a leaky barrier might be due to a faulty repair mechanism or the cells’ inability to detect leaks and flip the switch.”
The researchers are now working to determine how the switch gets flipped to turn on Rho at the right time and place and to identify other proteins that are part of the cellular repair crew to plug leaky biological barriers.
The team’s findings appear in the journal Developmental Cell. Additional coauthors are from the University of Michigan and the University of Edinburgh. The National Institutes of Health, National Science Foundation Graduate Research Fellowships, and a postdoctoral fellowship from the Japan Society for the Promotion of Science funded the research.
Source: University of Michigan



