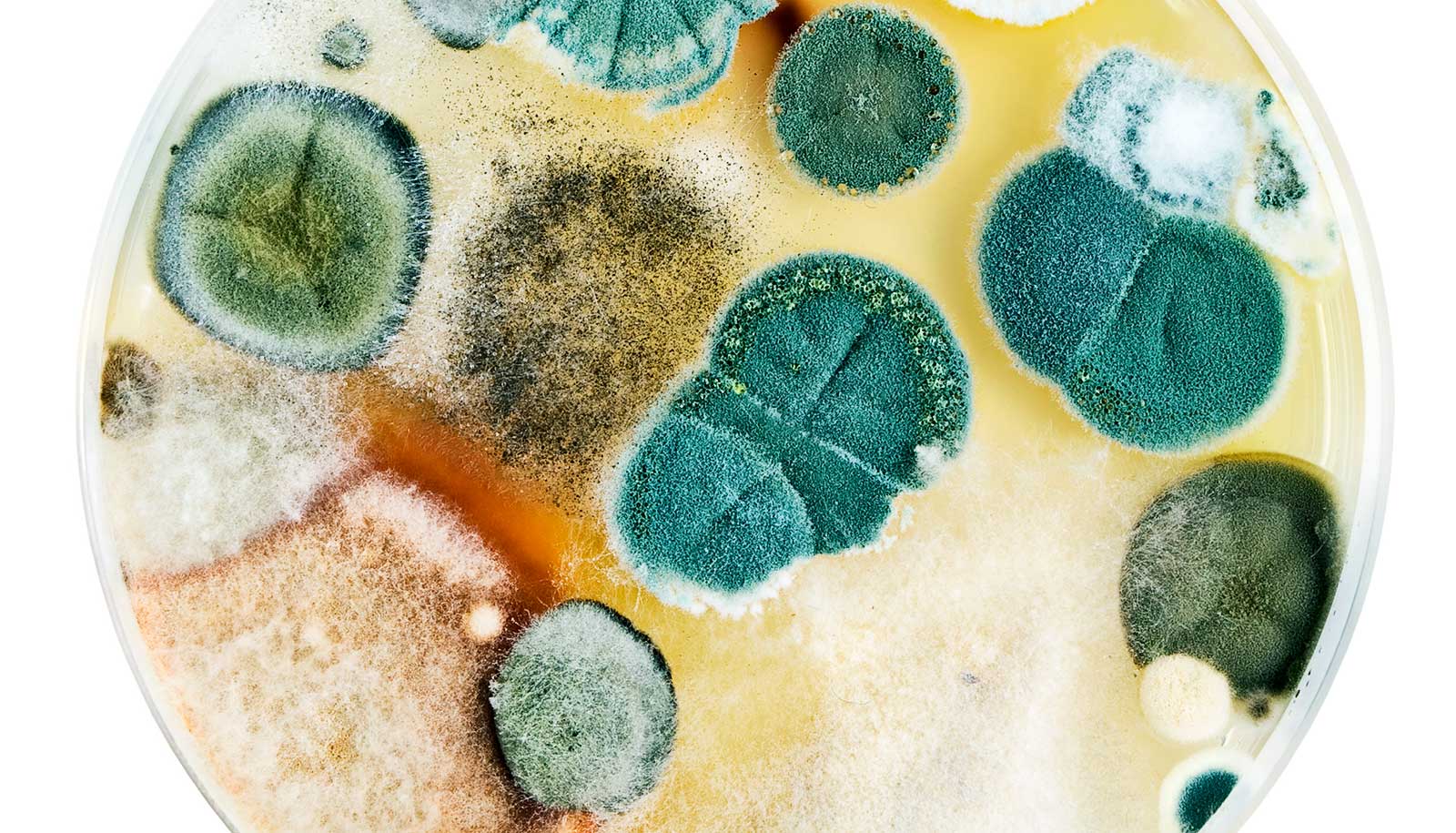
New technology uses genomics and data analytics to screen for molecules produced by molds to find potential new medication leads.
“…molds are a gold mine for new drugs…”
Fungi are rich sources of natural molecules for drug discovery, but numerous challenges have pushed pharmaceutical companies away from tapping into this bounty in recent years.
“Drug discovery needs to get back to nature, and molds are a gold mine for new drugs,” says Neil L. Kelleher, a chemical biologist at Northwestern University and director of the Proteomics Center of Excellence. “We have established a new platform that can be scaled for industry to provide a veritable fountain of new medicines. Instead of rediscovering penicillin, our method systematically pulls out valuable new chemicals and the genes that make them. They can then be studied in depth.”
Scientists believe there are thousands or even millions of fungal molecules waiting to be discovered, with enormous health, social, and economic benefits. The new technology systematically identifies powerful bioactive molecules from the microbial world—honed through millennia of evolution—for new drug leads.
These small molecules could lead to new antibiotics, immunosuppressant drugs, and treatments for high cholesterol, for example.
For four years, Kelleher has collaborated with Nancy P. Keller, a professor of mycology at the University of Wisconsin, and colleagues at Intact Genomics in St. Louis on developing the technology, called FAC-MS (Fungal Artificial Chromosomes with Metabolomic Scoring).
In recent work, the researchers applied their refined method to three diverse fungal species and discovered 17 new natural products from the 56 gene clusters they screened with the new process. That’s a hit rate of 30 percent, which, Kelleher says, is “absolutely phenomenal.”
“Fungi make these natural products for a reason, and a lot of them are antimicrobial,” says Keller, professor of medical microbiology and immunology and bacteriology at Wisconsin. “They’re used as weapons to kill or retard growth of other fungi, bacteria, or any other competing microbe in the area where the fungus wants to grow. Fungal compounds are a major source of diverse drugs.”
Each of the three institutions has played a key role in developing FAC-MS. The three-step system uses genomics and molecular biology to identify and capture large swaths of fungal DNA—called gene clusters—that are very likely to produce new molecules of interest, puts the DNA in a model fungus that grows easily in the lab, and then analyzes the chemical products using mass spectrometry and data analytics.
Scientists using fungal species for drug discovery have recently faced a number of problems: the slow rate at which researchers can systematically unlock fungal compounds; the rediscovery of old compounds, such as penicillin; the difference between what a fungus could produce versus what it actually does; and the ability to know when you have a new chemical as opposed to the thousands of more mundane compounds cells produce.
The team worked to address these problems to greatly increase the throughput of identifying new chemicals and the gene clusters responsible for their production.
‘Sieve’ sorts mirror-image molecules for new drugs
“Because these molecules are coming from a biological system, they tend to be more complex than a new molecule made in a pharmaceutical lab,” says Kenneth D. Clevenger, a fellow in Kelleher’s lab and a first author of the study.
“Molecules from fungi are predisposed to interact with cells and proteins, so, in that sense, they have promise. Our hope is that we find useful bioactivities that could lead to new medicines.”
The big advance, the researchers say, is how many gene clusters they were able to wrangle in a single study. Instead of reporting just one or two, they teed up 56 gene clusters and pulled out 17 new natural products and picked one to rigorously characterize in depth, which they named valactamide.
“We’ve designed a methodology where we can take all 56 gene clusters from fungi, package them and go through a process where we can try to express all of them,” says Jin Woo Bok, a senior scientist in Keller’s lab and a first author of the study.
How AI algorithms could help design new drugs
If brought to an industrial scale, the new FAC-MS process will help domesticate wild molds to reinvigorate drug discovery with compounds from the natural world.
The study appears in the journal Nature Chemical Biology. The National Institutes of Health supported the work.
Source: Northwestern University