A new “cryopreservation” technique opens a path to year-round sample collection, which is critical in managing threats to our oceans.
As ocean temperatures rise, coral reefs face increasing threats as ecosystem damage has accelerated in recent decades. The ability to preserve and revive biodiverse coral samples has become an essential priority for the health and future of the world’s oceans.
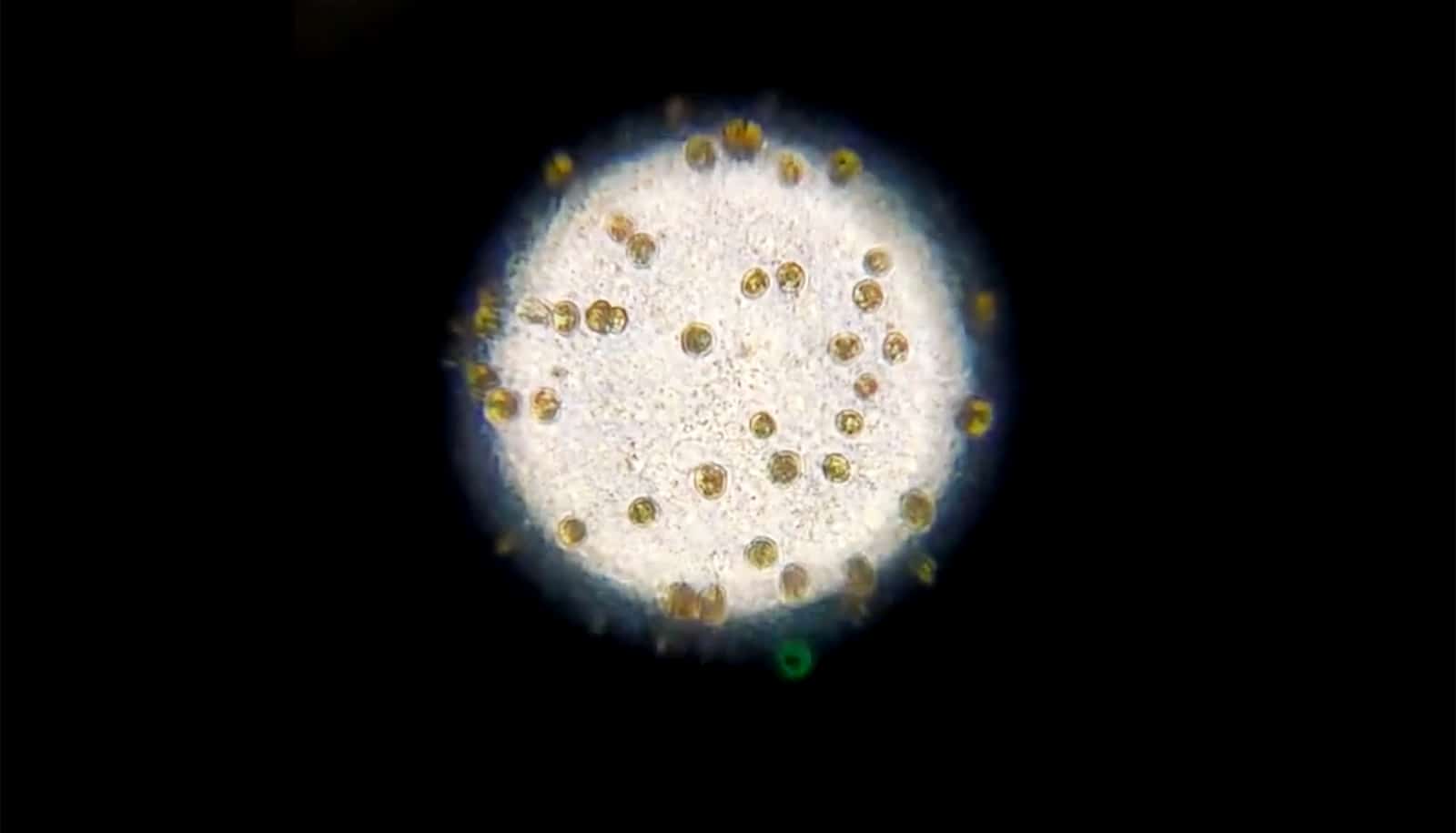
Now, researchers have demonstrated that live coral can be preserved through a new technique called “isochoric vitrification.” The new process allows the coral to be cooled rapidly to a temperature of -196 degrees Celsius—nearly -321 degrees Fahrenheit—without forming ice, which is lethal to coral and most biological matter.
The researchers took the technique into the real world, applying it to preserve coral fragments at the Hawaii Institute of Marine Biology.
Previous methods of cryopreservation involved collecting coral reproductive matter while the coral reproduced, which was limited to a few days out of the year and in often difficult-to-access locations.
Developing a technique not limited by the coral’s reproductive schedule makes it possible to extract and preserve coral throughout the year at any time.
To preserve the coral fragment without forming ice, the researchers first bleached the sample—a process in which a symbiotic photosynthetic algae is removed from the coral.
While in nature, bleaching is typically associated with overstress in coral and leads to starvation in the organism. However, researchers discovered that by applying isochoric vitrification without bleaching, the algae created ice pockets.
Once removed, the researchers could preserve the bleached coral fragment in a glassy state by introducing it to a chemical solution and storing it inside a specialized aluminum container that was then rapidly cooled using liquid nitrogen. The researchers then warmed and revived the coral by reintroducing filtered seawater gradually over 20 minutes.
While the technique is scientifically sophisticated, one of its greatest strengths is the simplicity of the technology required to apply it, says Matthew Powell-Palm, an assistant professor in the mechanical engineering department at Texas A&M University and lead author of the paper in Nature Communications.
“Compared to other emergent vitrification techniques—which frequently require lasers, electromagnetic implements, or other high-tech laboratory equipment—our isochoric vitrification approach is exceedingly fieldable,” Powell-Palm says.
“It requires no moving parts or electronics, and the protocol can be implemented by a field technician with no background in thermodynamics. This is essential to the practicality of any conservation technique because when this is deployed in real marine field stations, the high-tech lab infrastructure common to many laboratories will not be available. From a purely technological perspective, the technique is simple, rugged, and ready for the field.”
Looking forward, the researchers are working to reduce the stress placed on coral throughout the process. While the researchers successfully preserved and revived one species of coral, the life span observed was only up to 24 hours post-thaw. By reducing the overall stress placed on the coral, researchers hope to achieve long-term survival for subjects of the vitrification process and rapid deployment of the technique into real-world conservation efforts.
“Everyone benefits from the health of the world’s oceans, be it by eating fish, living in a coastal community that benefits from reef-driven tourism, or simply enjoying a walk on the beach,” Powell-Palm says. “Coral reefs are essential to the baseline health of our oceans, and cryo-conservation of endangered coral species can help to ensure that these invaluable and marvelous organisms do not go extinct.”
Coral reefs are among the planet’s most diverse ecosystems and are vital in ensuring food security, protecting coastlines, and serving as home to nearly a quarter of the ocean’s lifeforms.
Additional coauthors are from the University of California, Berkeley, and the Smithsonian’s National Zoo and Conservation Biology Institute.
The conservation organization Revive and Restore funded the work.
Source: Texas A&M University