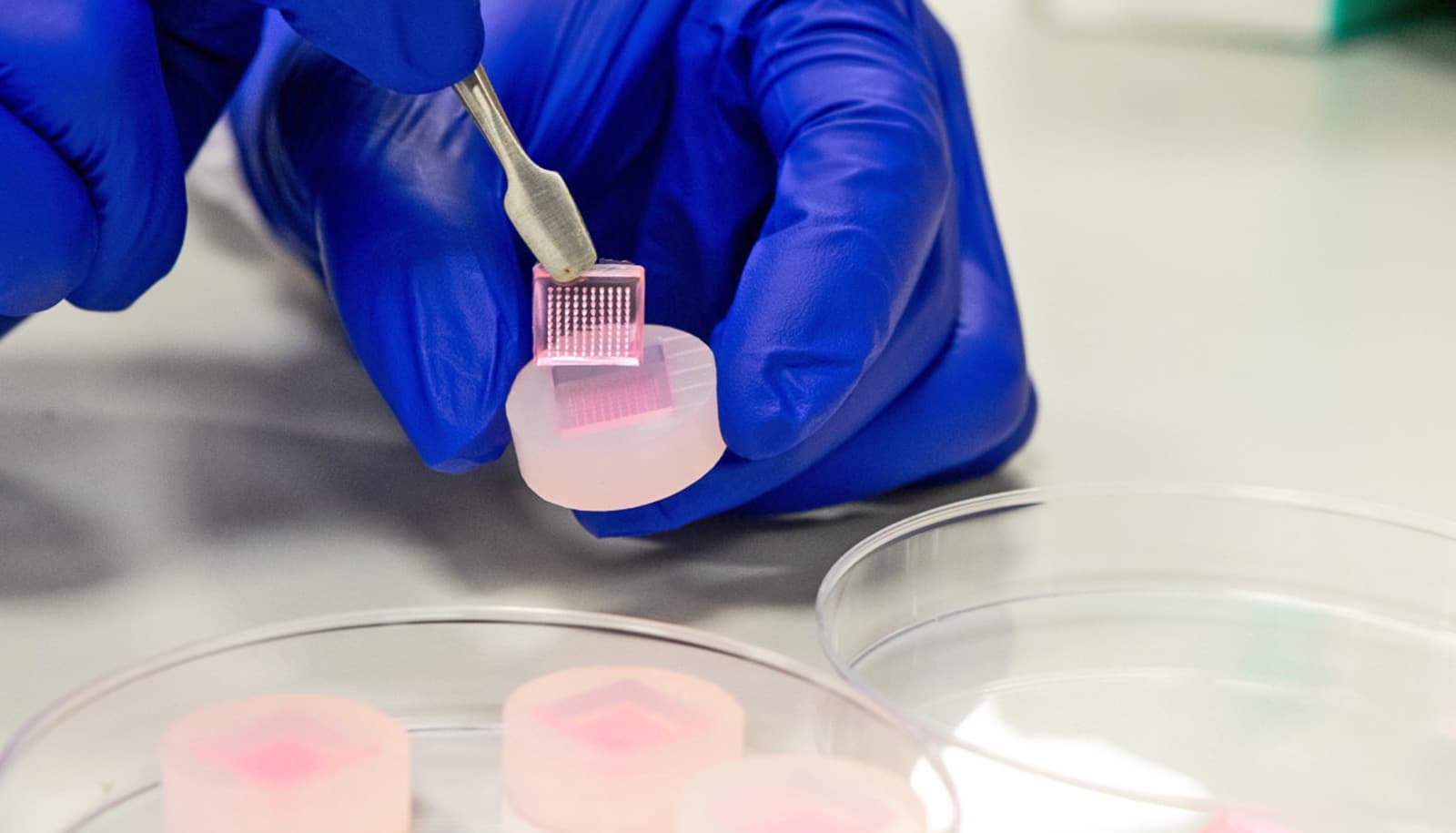
Researchers are working to develop a low-cost, practical biopolymer dressing that helps heal chronic wounds.
Tens of millions of patients around the world suffer from persistent and potentially life-threatening wounds. These chronic wounds, which are also a leading cause of amputation, have treatments, but the cost of existing wound dressings can prevent them from reaching people in need.
“The existing efficient technologies are far too expensive for most health care systems, greatly limiting their use in a timely manner,” says Morteza Mahmoudi, an assistant professor in the Michigan State University College of Human Medicine and the Precision Health Program. “An economically accessible, practical, and effective technology is needed.”
To develop that new technology, Mahmoudi tapped into his background studying advanced materials to heal heart tissue, fight infections, and support immune systems. But the team also kept an eye on cost, working to develop a product that could be made available to as many patients as possible, even in resource-constrained markets.
“My goal is always to make something that works and is practical,” Mahmoudi says. “I want to see my research become clinical products that help patients.”
As reported in the journal Molecular Pharmaceutics, Mahmoudi is getting closer to that goal. He’s working with partners in the United Kingdom who have started a company to oversee the development and approval of the new technology.
Working with his collaborators, Mahmoudi conducted a small pilot trial of the wound dressing with 13 patients with chronic wounds, all of whom were cured, he says.
Chronic wounds and diabetic food ulcers
Patients with advanced chronic wounds—those which do not respond to traditional therapies—are estimated to number over 45 million globally, making this one of the world’s most pressing and urgent health care needs, Mahmoudi says.
The United States is home to about 5% of this population, yet more than 90% of the sales of “active” wound care technologies happen in the US. That essentially means that the rest of the world is left out, Mahmoudi says.
Venous leg ulcers and pressure ulcers associated with immobility in older and paralyzed patients are also major causes of chronic wounds, but perhaps the best-known examples of this type of injury being diabetic foot ulcers. Worldwide, there are more than 400 million people living with diabetes, and some studies have estimated that up to a quarter of those patients will develop foot ulcers within their lifetime.
Even with the high level of care available in the US, more than 30% of patients who develop a diabetic foot ulcer will die within five years of its onset. For reference, that percentage is higher than breast cancer, prostate cancer, and colon cancer.
Diabetic foot ulcers also illustrate many of the reasons why chronic wounds can be so challenging to treat.
Patients with diabetes can be dealing with restricted blood flow and other factors that slow their immune response, compromising the body’s ability to heal the wound on its own. They can also have nerve damage that dulls the wound’s pain and can delay patients from seeking treatment. When wounds heal more slowly and stay open longer, bacteria have more opportunities to cause infections and lead to serious complications. Put bluntly, there’s a lot going wrong in a chronic wound.
Easy, practical, and cheap
“Chronic wounds are some of the most complicated things doctors have to treat,” Mahmoudi says. “If you want to make a dressing that works, it has to address all those problems. And in order to be relevant to the majority of patients in the world, it has to be easy to use, practical, and inexpensive as well.”
There are many technologies available to support healing in chronic wounds, but those that can stimulate tissue regeneration are typically derived from harvested natural tissues. This is complex and expensive, resulting in products that cost upwards of $1,000, putting them out of reach for many patients and health care systems.
By designing a product that can be manufactured from readily available biopolymers, production costs can be kept low, and the team could add various other materials to lead to improved healing.
Biopolymer design
The team starts with a flexible framework of nanofibers—exceedingly thin threads—of natural polymers, including collagen, a structural-support protein found in our skin and cartilage. The framework provides a three-dimensional scaffold that fosters cell migration and the development of new blood vessels, essentially replicating the function of the extracellular matrix, the natural support system found in healthy, living tissue.
“It’s important that the physical and mechanical properties of the dressing are really close to that of skin,” Mahmoudi says. “In order to heal, the new cells have to feel like they’re at home.”
To that framework, the team can incorporate proteins, peptides, and nanoparticles that not only spur the growth of new cells and blood vessels but also fight off bacteria by encouraging a patient’s own immune system to join the charge. (The team’s experiences on these elements appeared in earlier publications in Nature Nanotechnology and Trends in Biotechnology).
The dressing also degrades over time, meaning that nobody would have to change or remove it and potentially aggravate the wound site. And at roughly $20 apiece, Mahmoudi believes that the dressings—if and when regulatory agencies approve them—will be affordable to even resource-strapped health care systems faced with treating these serious wounds.
Collaborators on the project are from Harvard Medical School; Emory University; Georgia Tech; Rutgers State University; McGill University; University of Siegen in Germany; and the University of Santiago de Compostela in Spain.
Source: Matt Davenport for Michigan State University