Researchers have demonstrated that specialized nucleic acid-based nanostructures could target cancer cells while bypassing normal cells.
“Most of the therapeutic drugs are not able to discriminate the cancer cells from healthy cells…”
More than 100 years ago, German Nobel laureate Paul Ehrlich popularized the “magic bullet” concept—a method that clinicians might one day use to target invading microbes without harming other parts of the body. Although chemotherapies have been highly useful as targeted treatments for cancer, unwanted side effects still plague patients.
“Most of the therapeutic drugs are not able to discriminate the cancer cells from healthy cells,” says David Porciani, a postdoctoral fellow at the Bond Life Sciences Center at the University of Missouri.
“They are killing both cell populations (healthy and malignant), and the treatment can have harsher side effects than the cancer itself in the short term. We are developing ‘smart’ molecules that can bind with receptors that are found on the surface of cancer cells, thus representing a cancer signature,” Porciani says. “The idea is to use these smart molecules as vehicles to deliver chemotherapeutic drugs or diagnostics.”
Using a molecular process that mimics a highly-accelerated form of natural evolution, Porciani and his team sought out nucleic acid ligands, or aptamers. Because of their three-dimensional structures, researchers can train aptamers to bind to certain target molecules with high affinity and selectivity. When the target is a cancer-associated receptor, researchers can use these aptamers as molecular tools to recognize specifically diseased cells.
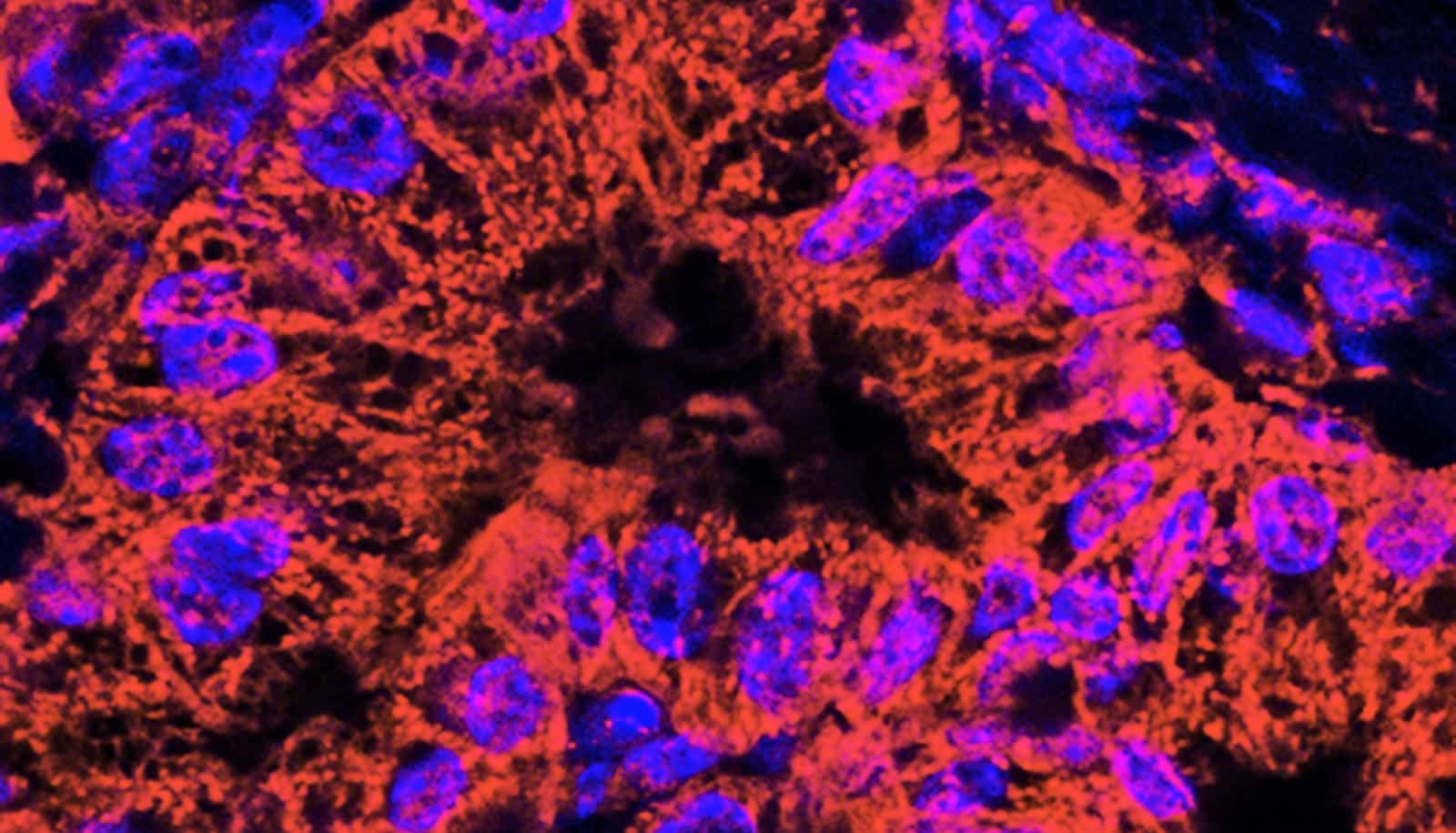
The team then “loaded” the aptamers with large, fluorescent RNAs generating nucleic-acid nanostructures. Upon incubation with target cancer and non-target cells, only malignant cells were illuminated by the nanostructure showing that the structures had correctly bonded with their intended targets.
Anti-cancer drug puts tumor cells to ‘sleep’
“Next steps for our studies are to prove that these aptamers can be loaded with therapeutic molecules that specifically target and treat cancer cells leaving normal tissues untouched,” Porciani says.
“While aptamers have been proven in the past as tools to deliver small drugs, our method paves the way to deliver even larger and potentially more powerful RNA-based drugs possibly creating that ‘magic bullet’ that Erhlich described in the last century.”
The study appears in Nature Communications. The National Institutes of Health provided funding. The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding agencies.
Source: University of Missouri